Spotlight on Targeted Chemotherapy in Retinoblastoma: Safety, Efficacy, and Patient Outcomes

Introduction
Retinoblastoma (RB) is a rare but the most common primary intraocular cancer in children.1 Inactivation of both copies of the RB1 gene, a tumor suppressor gene, as a main driver, in association with some other epigenetic alteration, is usually manifested by unifocal or multifocal tumors of retinal origin.2 Although historically RB was a lethal disease, with a survival rate less than 5% in the nineteenth century, nowadays the 5-year survival rate in developed countries is almost 100%.3 In spite of this progress, a striking disparity regarding the cure rate of RB and patient survival exists between developed and developing countries.4 While the disease is considered the most curable of all pediatric cancers in the developed world, usually it is associated with a high mortality rate, following dissemination and metastasis, in low-income nations.5,6 In an analysis of gross national income of the country versus RB mortality, Chantada et al7 reported that survival from RB is 30% in low-income countries, 60% in lower–middle-income countries, 75% in upper–middle-income countries, and 95% in high-income countries. In 2020, Gündüz et al8 reported the overall survival rate of 96% in an upper–middle-income country.
Although the first enucleation, by the Scottish surgeon James Wardrope,9 revolutionized the management of RB, by 1897, only 17% of children survived this deadly disease.10 One of the major turning points in the history of RB management, pioneered by Hilgartner11 in 1903 in Texas, and followed by Schoenberg,12 Verhoeff,13 and Moore et al,14 was radiotherapy in relatively small tumors; a remedy making eye salvage a possibility. In addition, the widespread application of focal therapies (eg photocoagulation and cryotherapy) for small RB, and the development of plaque radiotherapy by Stallard for larger unifocal tumors,15 enhanced prognosis in terms of both patient and globe survival.16
The introduction of systemic chemotherapy as the mainstay of treatment for primary RB using the VEC protocol (vincristine, etoposide, and carboplatin) in the last decade of the twentieth century turned RB into the most treatable cancer in the first decade of the twenty-first century.17–20 This was a great leap toward achieving patient survival of more than 90% and resulted in the change in the classification of RB from the Reese–Ellsworth grouping to the International Intraocular Retinoblastoma Classification (IIRC).21
As standard of care, systemic chemotherapy in children with intraocular RB averts the need for radiation and enucleation in many cases.22,23 Primary systemic chemotherapy was associated with excellent results in eye groups A–C (more than 90%) on the IIRC and a modest globe salvage rate (less than 50%) in group D, as well as a less favorable prognosis in group E (mostly enucleated) in the early days of the chemoreduction era;24 however, a report published in 2020, on the long-term (20-year) real-world outcomes of intravenous chemotherapy (IVC) in 964 eyes, revealed the tumor control rates for group A (96%), group B (91%), group C (91%), group D (71%), and group E (32%).25
A step forward for increased globe preservation was super-selective intra-arterial chemotherapy (IAC),26 which was based on the earlier experiences of Japanese clinicians who reported on selective ophthalmic arterial infusion (SOAI) therapy for RB.27 The published evidence indicates that IAC is more effective in providing globe retention in group D and E RB compared to IVC. With the delivery of a greater concentration of chemotherapeutic agents in the target tumor (direct delivery of 10 times more28) and less systemic toxicity, IAC came out as one of the first-line management options in RB, particularly in advanced unilateral group E and D cases.29,30 In spite of these advantages for IAC, it remains an invasive and costly procedure that needs to be undertaken by an experienced multidisciplinary team, which may not available in low- and even in some middle-income countries.
Vitreous seeding (VS) and subretinal seeding (SRS) were key obstacles to achieving globe retention in patients with intraocular RB. In fact, only a quarter of the eyes that developed VS following radiotherapy were salvaged prior to the advent of IAC and intravitreal chemotherapy (IVitC).31 Therefore, along with IVC, re-exploration of the forbidden therapeutic modality of IVitC in the 2010s, with the augmented benefit of controlling refractory VS, improved globe survival and made external beam radiotherapy (EBRT) a thing of the past.32
As a promising option for eye salvage, intracameral chemotherapy (IcamC) was introduced by Munier et al in 2017 for the treatment of anterior chamber (AC) and posterior chamber (PC) seeding (bicameral technique). Considering the poor penetration of chemotherapeutic agents to the AC by the systemic route, AC seeding was an indication for immediate enucleation prior to the era of this treatment modality.33
Nowadays, most advanced cases of RB are salvaged using targeted chemotherapy, without compromising the patient survival rates.31 Currently, over 90% of group D eyes and roughly half of group E eyes are saved, and excellent outcomes, associated with declining complication rates, have been achieved during the past decade,34 along with excellent patient survival.35,36
It should be noted that about 75% of affected eyes worldwide present with advanced RB, characterized by VS and SRS, as well as retinal detachment, in which IVC and radiation fail to result in globe preservation in the majority of cases. Moreover, the risk of secondary malignancies, fetal adverse effects, such as neutropenia, and ototoxicity associated with systemic chemotherapy, have convinced many ocular oncologists to choose targeted chemotherapy as the treatment of choice for their advanced cases.37
In this study, we aimed to present a comprehensive review of the treatment of intraocular RB with targeted chemotherapy, including intra-arterial chemotherapy, intravitreal chemotherapy, and intracameral chemotherapy. PubMed, Scopus, and Google Scholar were searched, with specific emphasis on articles published from 1990 to 2022. Retinoblastoma, intra-arterial chemotherapy, ophthalmic artery chemosurgery, intravitreal chemotherapy, and intracameral chemotherapy were the keywords used for the search terms.
Intra-Arterial Chemotherapy (Ophthalmic Artery Chemosurgery)
The history of IAC goes back to 1958, when Reese punctured the carotid artery to deliver nitrogen mustard directly to the eye. Although the tumor regressed initially, recurrence ensued and the patient was managed by radiotherapy. Years later, Japanese investigators revealed that melphalan is more effective against RB and they established selective IAC for the treatment of RB. Despite the Japanese experience with over 400 eyes, it was only adopted as the primary treatment for RB cases following the introduction of a modified technique by Abramson and Gobin at MSK in 2006.37 During IAC, the tip of the fluoroscopy-guided microcatheter is placed at the proximal part of the ophthalmic artery and typically one to three chemotherapy drugs (melphalan, topotecan, and carboplatin) are delivered. This treatment is typically provided every 3–4 weeks for a mean of three sessions.38
The microcatheter is positioned at the ostium of the ophthalmic artery and a selective angiogram with contrast is performed to verify the angioanatomy, placement, and choroidal blush, which indicates proper eye flow. In approximately 12.5% of all intra-arterial infusions, the ophthalmic artery cannot be catheterized for various reasons,39 including small size of the ophthalmic artery owing to young patient age, vasospasm, and alternative vascular supply to the eye, usually the middle meningeal artery (MMA), which is present in 1–5% of the population.40 Chemotherapeutics are diluted in 30 mL of normal saline and injected manually over 30 minutes in a pulsatile fashion to disrupt smooth flow and allow diffusion of drug to the supplied vascular territory.41
With the balloon technique, drugs are each diluted in 6 mL of normal saline and infused via the balloon over the 4-minute inflation time in order to limit the total number of balloon injection cycles to no more than three, if multifactorial treatment is performed.39
For group B and C eyes, melphalan is used alone at a dose of 3.5–5 mg, based on the patient’s weight. However, in advanced cases of group D and E disease, topotecan is added at a dose of 1 mg. Considering the severity of ocular toxicity caused by carboplatin (at a dose of 25 mg), this drug is seldom used except in tandem therapy of the fellow eye for the treatment of bilateral RB by IAC.42 It should also be noted that a limit of 6 kg for weight and 3–4 months for age exist for performing IAC, so for neonates and young infants “bridge chemotherapy” with systemic carboplatin (18.7 mg/kg IV every 3–4 weeks) is used until the patient’s bodily features permit the clinician to proceed to IAC.43
The targeted delivery of chemotherapeutics to the eye was confirmed by Taich et al, who recorded increased vitreous and retinal concentrations of topotecan following ophthalmic artery chemosurgery compared with intravenous administration.44 According to a rodent study, topotecan with carboplatin effectively stopped the progression of RB compared to vincristine, carboplatin, and etoposide.45 In healthy rabbits, melphalan concentrations were found to be 12-fold higher in the retina and 26-fold higher in the vitreous humor following IAC compared with intravenous delivery.46
In cases of RB, the presence of SRS makes it difficult to achieve a complete cure without enucleation; both focal and systemic chemotherapy are largely ineffective in such cases. However, the advent of IAC allowed physicians to salvage most of these eyes without compromising the survival of the patients.30 According to investigations on humans and animals, a “depot” delivery system is established in the space beneath the retina following intra-arterial drug administration, and the seeds are exposed to the chemotherapeutic agent for many hours.47
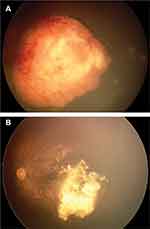
IAC has been used successfully both as an initial treatment and as an alternative modality for refractory group D and E cases.48 In addition, this modality can be used in refractory cases of groups A–C. Moreover, some experts believe that IAC alone can be the initial treatment of choice in all cases, regardless of IIRC grouping (Figure 1A and B).49 However, there are some concerns regarding IAC compared to IVC, including an increased chance of toxicity in the better eye, unknown effects on the prevention of pineoblastoma, and precluding the effects of systemic chemotherapy on potential pre-existing metastases.50
Before the era of IAC, group E eyes were routinely enucleated and the globe retention rate was around 47% in referral centers for group D.24 In comparison, with the introduction of IAC, enucleation was averted in up to half of eyes with group E disease, and globe survival improved to 82% in group D disease.25 Our experience showed similar outcomes; in the post-IAC era, the 17% and 1% globe survival rates in group D and E eyes were improved to 66% and 23%, respectively.51
Moreover, a 2021 meta-analysis has shown that the rate of globe survival for systemic chemotherapy alone is 93%, 83%, 73%, 40%, and 19% for group A–E eyes.52 In comparison, the 1-year Kaplan–Meier estimate for ocular survival after IAC was 96% across different groups.49 Table 1 shows the treatment outcomes of IAC for RB.
 |
 |
 |
Table 1 Treatment Outcomes of Intra-Arterial Chemotherapy for Retinoblastoma |
There are benefits associated with IAC in comparison to alternative treatments. By eliminating systemic multiagent chemotherapy, we can avert the necessity for a port, minimize the need for antimicrobial prophylaxis, and decrease the rate of iatrogenic neutropenia requiring blood transfusions. Chemotherapy-induced hair loss and carboplatin-induced deafness are also avoided. The rate of febrile neutropenia requiring a transfusion is less than 1% in IAC, and low CD4 counts are rarely encountered.53 As another advantage, secondary leukemia also seems to be averted, as this condition is yet to be reported in cases treated with IAC.36 Furthermore, the duration of therapy is shortened,6 and expenses are dramatically reduced since systemic chemotherapy requiring ports, antibiotics, hospitalization, and possible transfusions are eliminated. Routine vaccination schedules are also left undisrupted as the immune system is not affected as much as with systemic chemotherapy.53 In certain RB cases, choroidal invasion, orbital involvement, and optic nerve invasion have been treated successfully.54
IAC Safety Concerns
Considering the targeted delivery of chemotherapeutics to delicate retinal structures, despite averting the need for enucleation in most cases, the toxicity induced by IAC is a matter of concern. In this regard, a number of IAC-related complications have been reported.55 It is important to note that chemotherapeutics have fewer side effects and complications when used optimally.42
Transient grade III/IV neutropenia has been reported in 12% of patients; however, the risk of severe neutropenia increases at doses higher than 0.48 mg/kg.43,56 In fact, the rate of febrile neutropenia requiring a transfusion is less than 1% in IAC, and low CD4 counts are rarely encountered.53
In one study, the retina-related outcomes of IAC in patients with advanced RB and poor retinal function were studied via serial electroretinography under anesthesia58. Brodie et al investigated this issue in a small-scale study via serial electroretinography under anesthesia pre- and post-IAC in cases of advanced RB, revealing the persistence or even improvement of the function of the retina.57
In relation to the decrease in electroretinogram (ERG) response, melphalan and carboplatin have the strongest and weakest associations, respectively. In one study, multivariate analysis revealed the modest, short-term impact of melphalan on the amplitude change of the ERG, which could not be detected a year after the procedure.58 Although the toxicity varies across different chemotherapeutic agents, this variation is minimal when using the recommended dose of each drug and appears to have no clinical significance.58
Both the efficacy and cytotoxicity of melphalan depend on its dose and schedule. In a swine model, the post-IAC accumulation of this chemotherapeutic agent in the retinal pigment epithelium (RPE)–choroid was demonstrated. The researchers suggested that this phenomenon, which possibly explains the agent’s choroidal toxicity (in approximately 3–5% of patients) on the one hand and its efficacy in treating SRS on the other, could be due to the affinity of melphalan to melanin (Figure 2).59
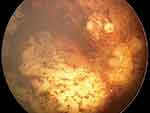 |
Figure 2 Extensive chorioretinal atrophy following intra-arterial chemotherapy. |
Other side effects vary depending on the chemotherapeutic agent used, and include palsy of the third cranial nerve, irreversible retinal detachment, vitreous hemorrhage, and restrictions in ocular motility; blindness may develop secondary to atrophy of the orbital fat and optic nerve and fibrosis of the latter.6,60 The rare possibility of central retinal artery occlusion is noted following IAC, though this complication appears to be strongly dependent on the experience of the treating team.38 Accelerated tumor regression of endophytic tumors may result in rhegmatogenous retinal detachment in 8–16% of cases treated with primary IAC.55
Periocular side effects including periorbital edema (in 10–15% of patients), and hyperemia, madarosis, ptosis, hair loss have also been reported following IAC. However, erythema usually resolves within a few months (Figure 1C).61 In rare instances, necrosis of periocular skin may ensue (Figure 3).
 |
Figure 3 Sequela of periocular skin necrosis following intra-arterial chemotherapy. (Written informed consent was obtained from the parents for the publication of the image). |
Femoral artery occlusion is another side effect. Although it may resolve within a week following treatment with aspirin,62 in some cases it can lead to catastrophic events and even limb amputation. Reversible cerebral vasoconstriction has also been reported, again without any permanent sequelae.63 Rarely, more severe side effects such as carotid artery dissection, stroke, and even death may occur following IAC.62
Hospitalization is not always required when intraoperative events are encountered, though refractory hypotension is one matter that should be managed carefully as it may signal an anaphylactoid reaction.64 In the case of treatment failure, the enucleated eye usually shows massive choroidal invasion on pathology, linked with a roughly 4% risk of extraocular relapse.65
Intravitreal Chemotherapy
IVitC was described in 2003 by Kaneko and Suzuki for refractory cases of VS. In this technique, to induce hypotonia, paracentesis of the AC is carried out prior to intravitreal injection of 20–30 µg melphalan or 20 µg of topotecan. In order to minimize the risk of extraocular extension, triple freeze–thaw cryotherapy is applied at the site of injection during needle withdrawal. Afterwards, the eye is wiggled to distribute the drug homogeneously within the eye.66 An alternative approach is “precision” IVitC, which was introduced in 2018. In this technique, the drug is delivered at a site near the localized VS under the guidance of indirect ophthalmoscopy. In contrast to the classic approach, the eye in not jiggled and the head is positioned to maximize the local drug delivery and minimize unwanted drug toxicity to the macula.67
In rats, a high retinal concentration of melphalan was recorded 15 minutes following its intravitreal administration,47 with this level being maintained 7 hours longer in the retina than in the vitreous humor, indicating the role of this therapy in cases of SRS. The IVitC route is suitable for treating diseases within the vitreous humor as a large proportion of the active drug is not impeded from reaching the target by ocular barriers and systemic metabolism. Although IVitC has been proved to be useful as an additional globe salvage therapy (Figure 4), choosing this modality as the initial therapy is not usually an accepted approach.
Among cases with VS, the initial globe retention rates were roughly 60%,68 rising later to around 80%. The addition of intravitreal melphalan to IAC led to a rise in the ocular salvage rate while minimizing the time to cure and total exposure to chemotherapy.69 More than nine-tenths of the group D eyes that were enucleated just over a decade ago are now spared, with no negative impact in terms of patient survival or extraocular tumor extension.70
In an attempt to control newly seen SRS in RB patients who had previously received IAC, Abramson et al reported successful results of intravitreal injections of melphalan and/or topotecan combined with 810 nm indirect laser (continuous wave). They found that these two modalities offer good safety and efficacy when used in combination, though the contribution of each to the success rate could not be clarified. The results were not associated with any adverse outcomes, such as extraocular extension, metastasis, or death.71
In a review published in 2021, it was shown that during the IVC era, the globe survival could be improved from 63% to 71% with IVitC, a result comparable to those achieved with IAC.72 In cases of VS, the efficacy of IVitC is 85–100%.70
Possible complications after IVitC include pupillary synechiae, iris atrophy, retinal vascular occlusion, cataract, optic atrophy, hypotony, phthisis bulbi, and vitreous hemorrhage.73 The treatment outcomes of IVitC for RB are shown in Table 2.
 |
Table 2 Treatment Outcomes of Intravitreal Chemotherapy for Retinoblastoma |
Electrophysiologic studies have revealed some degree of retinal toxicity and a 5.8 μV decrease in the ERG recording for every intravitreal injection of melphalan. This retinal damage may be intensified by concomitant IAC (within a week). In addition, this side effect is more pronounced in eyes with greater pigmentation (eyes with brown irides) owing to the greater take up of melphalan by melanin. The toxicity has been documented clinically as a salt-and-pepper retinopathy in the fundus because of the increased concentration of the drug in the RPE and choroid. Such retinal toxicity is not seen with intravitreal injection of topotecan.70,74 With regard to intravitreal carboplatin, there is a dearth of data regarding its efficacy and toxicity.75
In addition to posterior segment abnormalities, the toxic effect of melphalan on the anterior segment has been reported in small case series in the meridian of the injection, which may reflect the higher concentration of the drug.76
The safety of intravitreal injection can be increased by delivering the chemotherapeutic or biological agent to a tumor-free pars plana site using an antireflux approach with small 30–32-gauge needles.77 In a large-scale, worldwide study, no cases of extraocular extension were reported following intravitreal injection.78 One study revealed that in over 3000 intravitreal injections, extraocular extension was not seen at all, leading to a predicted rate of below 0.08%.78
Intracameral Chemotherapy
In 2017, Munier et al introduced IcamC as a means of achieving high drug concentrations within the AC.33 Prior to the advent of this technique, tumoricidal doses of chemotherapeutics could not be reached in this part of the eye, and eyes with aqueous seeding had to be enucleated or subjected to AC plaque radiotherapy.
It should be noted that during IcamC, the failure of drug delivery to possible PC seedings may result in recurrent AC seeding. Therefore, it is of utmost importance to inject the chemotherapeutic agent in both the AC and PC, as well as the epiciliary region.79 The agents used in IcamC are melphalan (15–20 μg/0.05 mL) and topotecan (7.5 μg/0.015 mL). The original technique involved complete paracentesis of aqueous humor from both AC and PC through a long small-gauge needle placed at the periphery of the cornea. Afterwards, the syringe of drug is replaced without withdrawing the needle. One-third of the chemotherapeutic agent is injected into the AC. and following the perforation of the iris root in a tumor-free region, the remaining two-thirds are directed toward the PC. After the injection has been completed, the needle removal site is subjected to cryotherapy.33 Performing ultrasound biomicroscopy prior to injection can help in visualizing the PC seeding and choosing a tumor-free site. In addition, the systemic suppression of aqueous humor by acetazolamide before the injection helps to impede the rapid turnover of the drug in the AC. Nevertheless, some authors only inject a small amount of the drug into the AC, with or without paracentesis.79
When combined with plaque radiotherapy, IcamC led to complete tumor control in a patient who was followed up for 3 years.80 One study found that IcamC and intravitreal therapy provided good efficacy in treating lesions of the ciliary body and AC.81 In a retrospective study by Munier et al, it was shown that IcamC was successful in the management of AC seeding in six out of 11 patients, with a mean number of 4.3 injections, and the disruption of the anterior hyaloid face was the most important risk factor for failure of globe preservation.82 Given the excellent short-term results, studies with longer periods of follow-up are currently being conducted.83
The adverse effects of this treatment modality include iris heterochromia and progressive cataract formation,82 though topotecan offers greater safety with equal efficacy compared with melphalan.28
Future Directions
A problem that may arise during the treatment of chemosensitive RBs is the development of cross-resistance. The mechanisms behind this phenomenon are complicated, possibly differing from those involved in resistance to individual drugs. Treatment failure is a devastating consequence of drug resistance, particularly in metastatic neoplasms.84
Novel strategies for managing RB include molecularly targeted therapies, tubulin-modifying molecules, immunotherapy, high-mobility group A (HMGA) protein, vitamin D analogs, angiogenesis inhabitation, neurotransmitter pathway disruption, arsenic trioxide, EDL-155, gene therapy, local drug delivery systems, new hydrogel implant, ncRNAs, aqueous humor markers, exosomes, and MLN4924 (pevonedistat). Advances in tumor biology in recent decades have led to the emergence of molecularly targeted therapies for RB. Some examples include MDMX-p53 response inhibitors (nutlin-3a), spleen tyrosine kinase (SYK) inhibitors, histone deacetylase (HDAC) inhibitors, and CEP1347 (a small-molecule kinase inhibitor).85,86 It is of note that many of these novel drugs can be administered locally. For example, a mouse model demonstrated the efficacy of subconjunctival nutlin-3A in reducing the RB tumor burden, particularly when used with topotecan (TPT), which is currently being trialed for treating RB.87,88
In addition, it seems that gene therapy holds a lot of promise for the treatment of RB.89 During suicide gene therapy, the genetic materials of viruses or bacteria are transferred into neoplastic cells with the aim of converting a non-toxic substance into a lethal drug. According to a phase I trial, the herpes simplex virus–tyrosine kinase (HSV-TK) adenovirus vector, along with ganciclovir (GCV), can safely and effectively treat VS via the intravitreal injection route. Inflammation is induced locally but not systemically during this therapy.90
Moreover, there are several novel approaches for the local delivery of drugs, which include dendrimers, liposomes, biodegradable polyesters, mesoporous silica, and gold nanoparticles. Specific cells can be targeted by these engineered particles.91 One agent that is particularly favored for ocular therapy is polylactic-co-glycolic acid (PLGA), which is well known for its biocompatibility, degradation properties, and clinical applications.92 Active drugs such as flurbiprofen have been delivered into the eye using PLGA NPs.93 Another injectable, biodegradable delivery system that is currently being tested is fibrin glue.94 Dendrimer macromolecules are synthetic polymers with a spherical shape, three domains, and diameters of 1–100 nm. Dendrimers have also been looked at in terms of their ophthalmologic drug delivery applications. The purpose of the novel hydrogel implant is to deliver chemotherapeutics directly to the globe.95
Another important aspect in the management of RB is the lack of tissue diagnosis in the case of globe-conserving therapies. Owing to the chance of extraocular extension of RB, direct tissue biopsy is prohibited. Recently, aqueous biopsy has been introduced as an alternative approach. Nowadays, RB can be managed without requiring enucleation as a result of the availability of aqueous humor markers and circulating tumor cell- and cfDNA-based fluid biopsies.96 High-resolution mass spectrometry can be used to detect exosomes in RB tumors and tumor seeding in the vitreous humor in RB cell lines. Hence, exosomal markers have emerged as potential markers for disease diagnosis, treatment monitoring, and evaluation of prognosis in RB.97
Conclusion
Although patient and globe survival have improved significantly with the implementation of targeted chemotherapy in the management of RB, along with mitigation of the systemic complications of chemotherapeutic agents and radiation, further development is needed to move toward vision-salvaging therapies. New studies have explored novel modalities such as drug delivery approaches, molecular specific targeted therapies, and new horizons in gene therapy to achieve better clinical outcome regarding conserving not only the globe but also, to some extent, vision, in advanced RB.86,98–101
The constellation of lessons learned from RB pathophysiology and the future development of targeted and less toxic therapies will make non-responder RB cases a thing of the past.101
Disclosure
The authors declare that they have no competing interests.
References
1. Luo C, Deng YP. Retinoblastoma: concerning its initiation and treatment. Int J Ophthalmol. 2013;6(3):397–401. doi:10.3980/j.issn.2222-3959.2013.03.26
2. Benavente CA, Dyer MA. Genetics and epigenetics of human retinoblastoma. Annu Rev Pathol. 2015;10:547–562. doi:10.1146/annurev-pathol-012414-040259
3. Chung CY, Medina CA, Aziz HA, Singh AD. Retinoblastoma: evidence for stage-based chemotherapy. Int Ophthalmol Clin. 2015 Winter;55(1):63–75. doi:10.1097/IIO.0000000000000054
4. Naseripour M. “Retinoblastoma survival disparity”: the expanding horizon in developing countries. Saudi J Ophthalmol. 2012;26(2):157–161. doi:10.1016/j.sjopt.2012.02.003
5. Fabian ID, Abdallah E, Abdullahi SU, et al. Global retinoblastoma presentation and analysis by national income level. JAMA oncolog. 2020;6(5):685–695. doi:10.1001/jamaoncol.2019.6716
6. Munier FL, Beck-Popovic M, Chantada GL, et al. Conservative management of retinoblastoma: challenging orthodoxy without compromising the state of metastatic grace. “Alive, with good vision and no comorbidity”. Prog Retin Eye Res. 2019;73:100764.
7. Chantada GL, Qaddoumi I, Canturk S, et al. Strategies to manage retinoblastoma in developing countries. Pediatr Blood Cancer. 2011;56(3):341–348. doi:10.1002/pbc.22843
8. Gündüz AK, Mirzayev I, Temel E, et al. A 20-year audit of retinoblastoma treatment outcomes. Eye. 2020;34(10):1916–1924. doi:10.1038/s41433-020-0898-9
9. Abramson DH. Retinoblastoma in the 20th century: past success and future challenges the Weisenfeld lecture. Invest Ophthalmol Vis Sci. 2005;46(8):2683–2691. doi:10.1167/iovs.04-1462
10. Albert DM. Historic review of retinoblastoma. Ophthalmology. 1987;94(6):654–662. doi:10.1016/s0161-6420(87)33407-4
11. Hilgartner HL. Report of case of double glioma treated with x-ray. 1903. Tex Med. 2005;101(7):10.
12. Schoenberg M. A case of bilateral glioma of the retina apparently arrested in the non-enucleated eye by radium treatment. Arch Ophthalmol. 1919;48:485–488.
13. Verhoeff FH. Glioma retinae treated by x-rays, with apparent destruction of the tumor and preservation of normal vision. Trans Am Ophthalmol Soc. 1921;19:209–216.
14. Moore RF, Stallard HB, Milner JG. Retinal gliomata treated by radon seeds. Br J Ophthalmol. 1931;15(12):673–696. doi:10.1136/bjo.15.12.673
15. Stallard HB. The treatment of retinoblastoma. Ophthalmologica. 1966;151(2):214–230. doi:10.1159/000304892
16. Shields JA. Diagnosis and management of intraocular tumors. CV Mosby; 1983.
17. Shields CL, De Potter P, Himelstein BP, Shields JA, Meadows AT, Maris JM. Chemoreduction in the initial management of intraocular retinoblastoma. Arch Ophthalmol. 1996;114(11):1330–1338. doi:10.1001/archopht.1996.01100140530002
18. Murphree AL, Villablanca JG, Deegan WF 3rd, et al. Chemotherapy plus local treatment in the management of intraocular retinoblastoma. Arch Ophthalmol. 1996;114(11):1348–1356. doi:10.1001/archopht.1996.01100140548005
19. Gallie BL, Budning A, DeBoer G, et al. Chemotherapy with focal therapy can cure intraocular retinoblastoma without radiotherapy. Arch Ophthalmol. 1996;114(11):1321–1328. doi:10.1001/archopht.1996.01100140521001
20. Kingston JE, Hungerford JL, Madreperla SA, Plowman PN. Results of combined chemotherapy and radiotherapy for advanced intraocular retinoblastoma. Arch Ophthalmol. 1996;114(11):1339–1343. doi:10.1001/archopht.1996.01100140539004
21. Linn Murphree A. Intraocular retinoblastoma: the case for a new group classification. Ophthalmol Clin North Am. 2005;18(1):41–53, viii. doi:10.1016/j.ohc.2004.11.003
22. Künkele A, Jurklies C, Wieland R, et al. Chemoreduction improves eye retention in patients with retinoblastoma: a report from the German Retinoblastoma Reference Centre. Br J Ophthalmol. 2013;97(10):1277–1283. doi:10.1136/bjophthalmol-2013-303452
23. Shields CL, Honavar SG, Meadows AT, et al. Chemoreduction plus focal therapy for retinoblastoma: factors predictive of need for treatment with external beam radiotherapy or enucleation. Am J Ophthalmol. 2002;133(5):657–664. doi:10.1016/S0002-9394(02)01348-X
24. Shields CL, Mashayekhi A, Au AK, et al. The international classification of retinoblastoma predicts chemoreduction success. Ophthalmology. 2006;113(12):2276–2280. doi:10.1016/j.ophtha.2006.06.018
25. Shields CL, Bas Z, Tadepalli S, et al. Long-term (20-year) real-world outcomes of intravenous chemotherapy (chemoreduction) for retinoblastoma in 964 eyes of 554 patients at a single centre. Br J Ophthalmol. 2020;104(11):1548–1555. doi:10.1136/bjophthalmol-2019-315572
26. Abramson DH. Super selective ophthalmic artery delivery of chemotherapy for intraocular retinoblastoma: ‘chemosurgery’ the first Stallard lecture. Br J Ophthalmol. 2010;94(4):396–399. doi:10.1136/bjo.2009.174268
27. Yamane T, Kaneko A, Mohri M. The technique of ophthalmic arterial infusion therapy for patients with intraocular retinoblastoma. Int J Clin Oncol. 2004;9(2):69–73. doi:10.1007/s10147-004-0392-6
28. Ancona-Lezama D, Dalvin LA, Shields CL. Modern treatment of retinoblastoma: a 2020 review. Indian J Ophthalmol. 2020;68(11):2356–2365. doi:10.4103/ijo.IJO_721_20
29. Shields CL, Manjandavida FP, Lally SE, et al. Intra-arterial chemotherapy for retinoblastoma in 70 eyes: outcomes based on the international classification of retinoblastoma. Ophthalmology. 2014;121(7):1453–1460. doi:10.1016/j.ophtha.2014.01.026
30. Abramson DH, Marr BP, Dunkel IJ, et al. Intra-arterial chemotherapy for retinoblastoma in eyes with vitreous and/or subretinal seeding: 2-year results. Br J Ophthalmol. 2012;96(4):499–502. doi:10.1136/bjophthalmol-2011-300498
31. Abramson DH. Retinoblastoma: saving life with vision. Annu Rev Med. 2014;65:171–184. doi:10.1146/annurev-med-061312-123455
32. Shields CL, Alset AE, Say EA, Caywood E, Jabbour P, Shields JA. Retinoblastoma control with primary intra-arterial chemotherapy: outcomes before and during the intravitreal chemotherapy era. J Pediatr Ophthalmol Strabismus. 2016;53(5):275–284. doi:10.3928/01913913-20160719-04
33. Munier FL, Gaillard MC, Decembrini S, Bongiovanni M, Beck-Popovic M. Intracameral chemotherapy (melphalan) for aqueous seeding in retinoblastoma: bicameral injection technique and related toxicity in a pilot case study. Ocul Oncol Pathol. 2017;3(2):149–155. doi:10.1159/000453617
34. Dalvin LA, Ancona-Lezama D, Lucio-Alvarez JA, Masoomian B, Jabbour P, Shields CL. Ophthalmic vascular events after primary unilateral intra-arterial chemotherapy for retinoblastoma in early and recent eras. Ophthalmology. 2018;125(11):1803–1811. doi:10.1016/j.ophtha.2018.05.013
35. Chen Q, Zhang B, Dong Y, et al. Comparison between intravenous chemotherapy and intra-arterial chemotherapy for retinoblastoma: a meta-analysis. BMC Cancer. 2018;18(1):486. doi:10.1186/s12885-018-4406-6
36. Habib LA, Francis JH, Fabius AW, Gobin PY, Dunkel IJ, Abramson DH. Second primary malignancies in retinoblastoma patients treated with intra-arterial chemotherapy: the first 10 years. Br J Ophthalmol. 2018;102(2):272–275. doi:10.1136/bjophthalmol-2017-310328
37. Schaiquevich P, Francis JH, Cancela MB, Carcaboso AM, Chantada GL, Abramson DH. Treatment of retinoblastoma: what is the latest and what is the future. Front Oncol. 2022;12:822330. doi:10.3389/fonc.2022.822330
38. Shields CL, Bianciotto CG, Jabbour P, et al. Intra-arterial chemotherapy for retinoblastoma: report No. 1, control of retinal tumors, subretinal seeds, and vitreous seeds. Arch Ophthalmol. 2011;129(11):1399–1406. doi:10.1001/archophthalmol.2011.150
39. Klufas M, Gobin Y, Marr B, Brodie S, Dunkel I, Abramson D. Intra-arterial chemotherapy as a treatment for intraocular retinoblastoma: alternatives to direct ophthalmic artery catheterization. Am J Neuroradiol. 2012;33(8):1608–1614. doi:10.3174/ajnr.A3019
40. Hayreh S. Orbital vascular anatomy. Eye. 2006;20(10):1130–1144. doi:10.1038/sj.eye.6702377
41. Gobin YP, Dunkel IJ, Marr BP, Brodie SE, Abramson DH. Intra-arterial chemotherapy for the management of retinoblastoma: four-year experience. Arch Ophthalmol. 2011;129(6):732–737. doi:10.1001/archophthalmol.2011.5
42. Manjandavida FP, Stathopoulos C, Zhang J, Honavar SG, Shields CL. Intra-arterial chemotherapy in retinoblastoma – A paradigm change. Indian J Ophthalmol. 2019;67(6):740–754. doi:10.4103/ijo.IJO_866_19
43. Gobin YP, Dunkel IJ, Marr BP, Francis JH, Brodie SE, Abramson DH. Combined, sequential intravenous and intra-arterial chemotherapy (bridge chemotherapy) for young infants with retinoblastoma. PLoS One. 2012;7(9):e44322. doi:10.1371/journal.pone.0044322
44. Taich P, Requejo F, Asprea M, et al. Topotecan delivery to the optic nerve after ophthalmic artery chemosurgery. PLoS One. 2016;11(3):e0151343. doi:10.1371/journal.pone.0151343
45. Laurie NA, Gray JK, Zhang J, et al. Topotecan combination chemotherapy in two new rodent models of retinoblastoma. Clin Cancer Res. 2005;11(20):7569–7578. doi:10.1158/1078-0432.CCR-05-0849
46. Daniels AB, Froehler MT, Kaczmarek JV, et al. Efficacy, toxicity, and pharmacokinetics of intra-arterial chemotherapy versus intravenous chemotherapy for retinoblastoma in animal models and patients. Transl Vis Sci Technol. 2021;10(11):10. doi:10.1167/tvst.10.11.10
47. Schaiquevich P, Fabius AW, Francis JH, Chantada GL, Abramson DH. Ocular pharmacology of chemotherapy for retinoblastoma. Retina. 2017;37(1):1–10. doi:10.1097/IAE.0000000000001275
48. Abramson DH, Daniels AB, Marr BP, et al. Intra-arterial chemotherapy (ophthalmic artery chemosurgery) for group D retinoblastoma. PLoS One. 2016;11(1):e0146582. doi:10.1371/journal.pone.0146582
49. Francis JH, Levin AM, Zabor EC, Gobin YP, Abramson DH. Ten-year experience with ophthalmic artery chemosurgery: ocular and recurrence-free survival. PLoS One. 2018;13(5):e0197081. doi:10.1371/journal.pone.0197081
50. Shields CL, Lally SE, Leahey AM, et al. Targeted retinoblastoma management: when to use intravenous, intra-arterial, periocular, and intravitreal chemotherapy. Curr Opin Ophthalmol. 2014;25(5):374–385. doi:10.1097/ICU.0000000000000091
51. Naseripour M, Sedaghat A, Mirshahi R, et al. Targeted chemotherapy in retinoblastoma: a step forward from patient survival to globe survival. Eur J Ophthalmol. 2022:11206721221093018. doi:10.1177/11206721221093018
52. Daniels AB, Patel SN, Milam RW, Kohanim S, Friedman DL, Koyama T. Effect of intravenous chemotherapy regimen on globe salvage success rates for retinoblastoma based on disease class-a meta-analysis. Cancers. 2021;13(9):2216. doi:10.3390/cancers13092216
53. Fischer C, Petriccione M, Vitolano S, Guarini E, Davis ME, Dunkel IJ. The effect of ophthalmic artery chemosurgery on immune function in retinoblastoma patients: a single institution retrospective analysis. J Pediatr Hematol Oncol. 2017;39(7):555–559. doi:10.1097/mph.0000000000000968
54. Abramson DH, Gobin YP, Dunkel IJ, Francis JH. Successful treatment of massive choroidal invasion in retinoblastoma with intra-arterial chemotherapy (ophthalmic artery chemosurgery). Ophthalmol Retina. 2021;5(9):936–939. doi:10.1016/j.oret.2020.12.018
55. Vajzovic LM, Murray TG, Aziz-Sultan MA, et al. Supraselective intra-arterial chemotherapy: evaluation of treatment-related complications in advanced retinoblastoma. Clin Ophthalmol. 2011;5:171.
56. Dunkel IJ, Shi W, Salvaggio K, et al. Risk factors for severe neutropenia following intra-arterial chemotherapy for intra-ocular retinoblastoma. PLoS One. 2014;9(10):e108692. doi:10.1371/journal.pone.0108692
57. Brodie SE, Pierre Gobin Y, Dunkel IJ, Kim JW, Abramson DH. Persistence of retinal function after selective ophthalmic artery chemotherapy infusion for retinoblastoma. Doc. Ophthalmol Proc. 2009;119(1):13–22. doi:10.1007/s10633-008-9164-3
58. Francis JH, Abramson DH, Gobin YP, et al. Electroretinogram monitoring of dose-dependent toxicity after ophthalmic artery chemosurgery in retinoblastoma eyes: six year review. PLoS One. 2014;9(1):e84247. doi:10.1371/journal.pone.0084247
59. Schaiquevich P, Buitrago E, Taich P, et al. Pharmacokinetic analysis of melphalan after superselective ophthalmic artery infusion in preclinical models and retinoblastoma patients. Invest Ophthalmol Vis Sci. 2012;53(7):4205–4212. doi:10.1167/iovs.12-9501
60. Schmack I, Hubbard GB, Kang SJ, Aaberg JTM, Grossniklaus HE. Ischemic necrosis and atrophy of the optic nerve after periocular carboplatin injection for intraocular retinoblastoma. Am J Ophthalmol. 2006;142(2):310–315. e2. doi:10.1016/j.ajo.2006.02.044
61. Marr B, Gobin PY, Dunkel IJ, Brodie SE, Abramson DH. Spontaneously resolving periocular erythema and ciliary madarosis following intra-arterial chemotherapy for retinoblastoma. Middle East Afr J Ophthalmol. 2010;17(3):207. doi:10.4103/0974-9233.65492
62. Sarici A, Kizilkilic O, Celkan T, Gode S. Blue toe syndrome as a complication of intra-arterial chemotherapy for retinoblastoma. JAMA Ophthalmol. 2013;131(6):801–802. doi:10.1001/jamaophthalmol.2013.1458
63. Abruzzo T, Patino M, Leach J, Rahme R, Geller J. Cerebral vasoconstriction triggered by sympathomimetic drugs during intra-atrerial chemotherapy. Pediatr Neurol. 2013;48(2):139–142. doi:10.1016/j.pediatrneurol.2012.10.005
64. Scharoun JH, Han JH, Gobin YP. Anesthesia for ophthalmic artery chemosurgery. Anesthesiology. 2017;126(1):165–172. doi:10.1097/aln.0000000000001381
65. Bosaleh A, Sampor C, Solernou V, et al. Outcome of children with retinoblastoma and isolated choroidal invasion. Arch Ophthalmol. 2012;130(6):724–729. doi:10.1001/archophthalmol.2012.567
66. Kaneko A, Suzuki S. Eye-preservation treatment of retinoblastoma with vitreous seeding. Jpn J Clin Oncol. 2003;33(12):601–607. doi:10.1093/jjco/hyg113
67. Yu MD, Dalvin LA, Welch RJ, Shields CL. Precision Intravitreal Chemotherapy for Localized Vitreous Seeding of Retinoblastoma. Ocul Oncol Pathol. 2019;5(4):284–289. doi:10.1159/000491432
68. Abramson DH, Shields CL, Munier FL, Chantada GL. Treatment of retinoblastoma in 2015: agreement and disagreement. JAMA Ophthalmol. 2015;133(11):1341–1347. doi:10.1001/jamaophthalmol.2015.3108
69. Scelfo C, Francis JH, Khetan V, et al. An international survey of classification and treatment choices for group D retinoblastoma. Int J Ophthalmol. 2017;10(6):961–967. doi:10.18240/ijo.2017.06.20
70. Francis JH, Brodie SE, Marr B, Zabor EC, Mondesire-Crump I, Abramson DH. Efficacy and toxicity of intravitreous chemotherapy for retinoblastoma: four-year experience. Ophthalmology. 2017;124(4):488–495. doi:10.1016/j.ophtha.2016.12.015
71. Abramson DH, Catalanotti F, Brodie SE, Kellick MG, Francis JH. Intravitreal chemotherapy and laser for newly visible subretinal seeds in retinoblastoma. Ophthalmic Genet. 2018;39(3):353–356. doi:10.1080/13816810.2018.1443343
72. Raval V, Bowen RC, Soto H, Singh A. Intravenous chemotherapy for retinoblastoma in the era of intravitreal chemotherapy: a systematic review. Ocul Oncol Pathol. 2021;7(2):142–148. doi:10.1159/000510506
73. Xue K, Ren H, Meng F, Zhang R, Qian J. Ocular toxicity of intravitreal melphalan for retinoblastoma in Chinese patients. BMC Ophthalmol. 2019;19(1):1–8. doi:10.1186/s12886-019-1059-4
74. Francis JH, Schaiquevich P, Buitrago E, et al. Local and systemic toxicity of intravitreal melphalan for vitreous seeding in retinoblastoma: a preclinical and clinical study. Ophthalmology. 2014;121(9):1810–1817. doi:10.1016/j.ophtha.2014.03.028
75. Smith SJ, Pulido JS, Salomão DR, Smith BD, Mohney B. Combined intravitreal and subconjunctival carboplatin for retinoblastoma with vitreous seeds. Br J Ophthalmol. 2012;96(8):1073–1077. doi:10.1136/bjophthalmol-2011-300829
76. Francis JH, Marr BP, Brodie SE, Abramson DH. Anterior ocular toxicity of intravitreous melphalan for retinoblastoma. JAMA Ophthalmol. 2015;133(12):1459–1463. doi:10.1001/jamaophthalmol.2015.3119
77. Munier FL, Gaillard M-C, Balmer A, et al. Intravitreal chemotherapy for vitreous disease in retinoblastoma revisited: from prohibition to conditional indications. Br J Ophthalmol. 2012;96(8):1078–1083. doi:10.1136/bjophthalmol-2011-301450
78. Francis JH, Abramson DH, Ji X, et al. Risk of extraocular extension in eyes with retinoblastoma receiving intravitreous chemotherapy. JAMA Ophthalmol. 2017;135(12):1426–1429. doi:10.1001/jamaophthalmol.2017.4600
79. Kaliki S. Aqueous seeding in intraocular retinoblastoma: a review. Clin Exp Ophthalmol. 2021;49(6):606–614. doi:10.1111/ceo.13964
80. Paez-Escamilla M, Bagheri N, Teira LE, Corrales-Medina FF, Harbour W. Intracameral topotecan hydrochloride for anterior chamber seeding of retinoblastoma. JAMA Ophthalmol. 2017;135(12):1453–1454. doi:10.1001/jamaophthalmol.2017.4603
81. Cassoux N, Aerts I, Lumbroso-Le Rouic L, Freneaux P, Desjardins L. Eye salvage with combination of intravitreal and intracameral melphalan injection for recurrent retinoblastoma with anterior chamber involvement: report of a case. Ocul Oncol Pathol. 2017;3(2):129–132. doi:10.1159/000452305
82. Munier FL, Moulin A, Gaillard MC, et al. Intracameral chemotherapy for globe salvage in retinoblastoma with secondary anterior chamber invasion. Ophthalmology. 2018;125(4):615–617. doi:10.1016/j.ophtha.2017.11.010
83. Borroni D, Bonzano C, Rachwani-Anil R, et al. Less is more: new one-step intracameral chemotherapy technique. Rom J Ophthalmol. 2021;65(3):218–221. doi:10.22336/rjo.2021.44
84. Nishida, S, Tsubaki, M. 誌上ンポジウムがん剤耐性克服を目指した分子標的の探索と治療法の開発西田升三椿正寛 [Exploration of Molecular Targets in the Development of New Therapeutics Aimed at Overcoming Multidrug Resistance]. Yakugaku zasshi: Journal of the Pharmaceutical Society of Japan. 2017;137(2):145–149. Japanese. doi:10.1248/yakushi.16-00229-5
85. Pritchard EM, Dyer MA, Guy RK. Progress in small molecule therapeutics for the treatment of retinoblastoma. Mini Rev Med Chem. 2016;16(6):430–454. doi:10.2174/1389557515666150722100610
86. Togashi K, Okada M, Suzuki S, et al. Inhibition of retinoblastoma cell growth by CEP1347 through activation of the P53 pathway. Anticancer Res. 2020;40(9):4961–4968. doi:10.21873/anticanres.14499
87. Brennan RC, Federico S, Bradley C, et al. Targeting the p53 pathway in retinoblastoma with subconjunctival Nutlin-3a. Cancer Res. 2011;71(12):4205–4213. doi:10.1158/0008-5472.can-11-0058
88. Laurie NA, Donovan SL, Shih CS, et al. Inactivation of the p53 pathway in retinoblastoma. Nature. 2006;444(7115):61–66. doi:10.1038/nature05194
89. Kaji EH, Leiden JM. Gene and stem cell therapies. JAMA. 2001;285(5):545–550. doi:10.1001/jama.285.5.545
90. Chévez-Barrios P, Chintagumpala M, Mieler W, et al. Response of retinoblastoma with vitreous tumor seeding to adenovirus-mediated delivery of thymidine kinase followed by ganciclovir. J Clin Oncol. 2005;23(31):7927–7935. doi:10.1200/jco.2004.00.1883
91. Bertrand N, Wu J, Xu X, Kamaly N, Farokhzad OC. Cancer nanotechnology: the impact of passive and active targeting in the era of modern cancer biology. Adv Drug Deliv Rev. 2014;66:2–25. doi:10.1016/j.addr.2013.11.009
92. You S, Luo J, Grossniklaus HE, Gou ML, Meng K, Zhang Q. Nanomedicine in the application of uveal melanoma. Int J Ophthalmol. 2016;9(8):1215–1225. doi:10.18240/ijo.2016.08.20
93. Weng Y, Liu J, Jin S, Guo W, Liang X, Hu Z. Nanotechnology-based strategies for treatment of ocular disease. Acta Pharm Sin B. 2017;7(3):281–291. doi:10.1016/j.apsb.2016.09.001
94. Martin NE, Kim JW, Abramson DH. Fibrin sealant for retinoblastoma: where are we? J Ocul Pharmacol Ther. 2008;24(5):433–438. doi:10.1089/jop.2007.0110
95. Yousef YA, Halliday W, Chan HS, Héon E, Gallie BL, Dimaras H. No ocular motility complications after subtenon topotecan with fibrin sealant for retinoblastoma. Can J Ophthalmol. 2013;48(6):524–528. doi:10.1016/j.jcjo.2013.05.018
96. Tarazona N, Cervantes A. Liquid biopsy: another tool towards tailored therapy in colorectal cancer. Ann Oncol. 2018;29(1):7–8. doi:10.1093/annonc/mdx641
97. Galardi A, Colletti M, Lavarello C, et al. Proteomic profiling of retinoblastoma-derived exosomes reveals potential biomarkers of vitreous seeding. Cancers. 2020;12(6):1555. doi:10.3390/cancers12061555
98. Zhang J, Benavente CA, McEvoy J, et al. A novel retinoblastoma therapy from genomic and epigenetic analyses. Nature. 2012;481(7381):329–334. doi:10.1038/nature10733
99. Arshad R, Barani M, Rahdar A, et al. Multi-functionalized nanomaterials and nanoparticles for diagnosis and treatment of retinoblastoma. Biosensors. 2021;11(4). doi:10.3390/bios11040097
100. Zhang Y, Duan S, Jang A, Mao L, Liu X, Huang G. JQ1, a selective inhibitor of BRD4, suppresses retinoblastoma cell growth by inducing cell cycle arrest and apoptosis. Exp Eye Res. 2021;202:108304. doi:10.1016/j.exer.2020.108304
101. Azimi F, Mirshahi R, Naseripour M. New horizons in retinoblastoma treatment: an updated review article. Mol Vis. 2022;28:130–146.
102. Marr BP, Hung C, Gobin YP, Dunkel IJ, Brodie SE, Abramson DH. Success of intra-arterial chemotherapy (chemosurgery) for retinoblastoma: effect of orbitovascular anatomy. Arch Ophthalmol. 2012;130(2):180–185. doi:10.1001/archophthalmol.2011.386
103. Parareda A, Català J, Carcaboso AM, et al. Intra‐arterial chemotherapy for retinoblastoma. Challenges of a prospective study. Acta Ophthalmol. 2014;92(3):209–215. doi:10.1111/aos.12295
104. Tuncer S, Sencer S, Kebudi R, Tanyıldız B, Cebeci Z, Aydın K. Superselective intra‐arterial chemotherapy in the primary management of advanced intra‐ocular retinoblastoma: first 4‐year experience from a single institution in Turkey. Acta Ophthalmol. 2016;94(7):e644–e651. doi:10.1111/aos.13077
105. Kiratli H, Koç İ, Inam O, Varan A, Akyüz C. Retrospective analysis of primarily treated group D retinoblastoma. Graefes Arch Clin Exp Ophthalmol. 2018;256(11):2225–2231. doi:10.1007/s00417-018-4051-4
106. Dalvin LA, Kumari M, Essuman VA, et al. Primary intra-arterial chemotherapy for retinoblastoma in the intravitreal chemotherapy era: five years of experience. Ocul Oncol Pathol. 2019;5(2):139–146. doi:10.1159/000491580
107. Peterson EC, Elhammady MS, Quintero-Wolfe S, Murray TG, Aziz-Sultan MA. Selective ophthalmic artery infusion of chemotherapy for advanced intraocular retinoblastoma: initial experience with 17 tumors. J Neurosurg. 2011;114(6):1603–1608. doi:10.3171/2011.1.JNS10466
108. Muen WJ, Kingston JE, Robertson F, Brew S, Sagoo MS, Reddy MA. Efficacy and complications of super-selective intra-ophthalmic artery melphalan for the treatment of refractory retinoblastoma. Ophthalmology. 2012;119(3):611–616. doi:10.1016/j.ophtha.2011.08.045
109. Leal-Leal CA, Asencio-López L, Higuera-Calleja J, et al. Globe salvage with intra-arterial topotecan-melphalan chemotherapy in children with a single eye. Rev Invest Clin. 2016;68(3):137–142.
110. Hua J, Gang S, Yizhou J, Jing Z. Intra-arterial chemotherapy as second-line treatment for advanced retinoblastoma: a 2-year single-center study in China. J Cancer Res Ther. 2018;14(1):106. doi:10.4103/jcrt.JCRT_722_17
111. Lee DH, Han JW, Hahn SM, et al. Changes in treatment patterns and globe salvage rate of advanced retinoblastoma in Korea: efficacy of intra-arterial chemotherapy. J Clin Med. 2021;10(22):5421. doi:10.3390/jcm10225421
112. Ghassemi F, Dehghani S, Mahmoudzadeh R, Khodabandeh A, Ghanaati H, Termehchi G. Five-year experience in treatment of retinoblastoma with intra-arterial chemotherapy: a single-center analysis. J Curr Ophthalmol. 2021;33(4):468. doi:10.4103/joco.joco_113_21
113. Suzuki S, Yamane T, Mohri M, Kaneko A. Selective ophthalmic arterial injection therapy for intraocular retinoblastoma: the long-term prognosis. Ophthalmology. 2011;118(10):2081–2087. doi:10.1016/j.ophtha.2011.03.013
114. Thampi S, Hetts SW, Cooke DL, et al. Superselective intra-arterial melphalan therapy for newly diagnosed and refractory retinoblastoma: results from a single institution. Clin Ophthalmol. 2013;7:981. doi:10.2147/OPTH.S43398
115. Ghassemi F, Shields CL, Ghadimi H, Khodabandeh A, Roohipoor R. Combined intravitreal melphalan and topotecan for refractory or recurrent vitreous seeding from retinoblastoma. JAMA Ophthalmol. 2014;132(8):936–941. doi:10.1001/jamaophthalmol.2014.414
116. Ong SJ, Chao A-N, Wong H-F, Liou K-L, Kao L-Y. Selective ophthalmic arterial injection of melphalan for intraocular retinoblastoma: a 4-year review. Jpn J Ophthalmol. 2015;59(2):109–117. doi:10.1007/s10384-014-0356-y
117. Chen M, Zhao J, Xia J, et al. Intra-arterial chemotherapy as primary therapy for retinoblastoma in infants less than 3 months of age: a series of 10 case-studies. PLoS One. 2016;11(8):e0160873. doi:10.1371/journal.pone.0160873
118. Michaels ST, Abruzzo TA, Augsburger JJ, Corrêa ZM, Lane A, Geller JI. Selective ophthalmic artery infusion chemotherapy for advanced intraocular retinoblastoma: CCHMC early experience. J Pediatr Hematol Oncol. 2016;38(1):65–69. doi:10.1097/MPH.0000000000000471
119. Chen M, Jiang H, Zhang J, et al. Outcome of intra‐arterial chemotherapy for retinoblastoma and its influencing factors: a retrospective study. Acta Ophthalmol. 2017;95(6):613–618. doi:10.1111/aos.13333
120. Munier FL, Mosimann P, Puccinelli F, et al. First-line intra-arterial versus intravenous chemotherapy in unilateral sporadic group D retinoblastoma: evidence of better visual outcomes, ocular survival and shorter time to success with intra-arterial delivery from retrospective review of 20 years of treatment. Br J Ophthalmol. 2017;101(8):1086–1093.
121. Rishi P, Sharma T, Sharma M, et al. Intra-arterial chemotherapy for retinoblastoma: two-year results from tertiary eye-care center in India. Indian J Ophthalmol. 2017;65(4):311. doi:10.4103/ijo.IJO_843_16
122. Francis JH, Roosipu N, Levin AM, et al. Current treatment of bilateral retinoblastoma: the impact of intraarterial and intravitreous chemotherapy. Neoplasia. 2018;20(8):757–763. doi:10.1016/j.neo.2018.05.007
123. Funes S, Sampor C, Villasante F, et al. Feasibility and results of an intraarterial chemotherapy program for the conservative treatment of retinoblastoma in Argentina. Pediatr Blood Cancer. 2018;65(8):e27086. doi:10.1002/pbc.27086
124. Rojanaporn D, Chanthanaphak E, Boonyaopas R, Sujirakul T, Hongeng S, Ayudhaya SSN. Intra-arterial chemotherapy for retinoblastoma: 8-year experience from a tertiary referral institute in Thailand. Asia Pac J Ophthalmol. 2019;8(3):211–217.
125. Liu CC, Mohmood A, Hamzah N, Lau JH, Khaliddin N, Rahmat J. Intra-arterial chemotherapy for retinoblastoma: our first three-and-a-half years’ experience in Malaysia. PLoS One. 2020;15(5):e0232249. doi:10.1371/journal.pone.0232249
126. Rishi P, Agarwal A, Chatterjee P, et al. Intra-arterial chemotherapy for retinoblastoma: four-year results from tertiary center in India. Ocul Oncol Pathol. 2020;6(1):66–73. doi:10.1159/000500010
127. Shields CL, Dockery PW, Yaghy A, et al. Intra-arterial chemotherapy for retinoblastoma in 341 consecutive eyes (1292 infusions): comparative analysis of outcomes based on patient age, race, and sex. J Am Ass Pediatric Pediatr Ophthalmol Strabismus. 2021;25(3):150.e1–150. e9. doi:10.1016/j.jaapos.2020.12.006
128. Kiefer T, Schlüter S, Bechrakis NE, et al. Intraarterial chemotherapy for retinoblastoma–initial experiences of a German reference centre. Klin Monatsblatter fur Augenheilkd. 2021;238(07):788–796. doi:10.1055/a-1508-6194
129. Mirzayev I, Gündüz AK, Yavuz K, et al. Secondary intra-arterial chemotherapy and/or intravitreal chemotherapy as salvage treatment for retinoblastoma. Eur J Ophthalmol. 2021;31(5):2692–2698. doi:10.1177/1120672120957587
130. Li J, Jing C, Hua X, et al. Outcome of salvage intra-arterial chemotherapy for recurrent retinoblastoma. Eye. 2021;36:1–5.
131. Berry JL, Shah S, Bechtold M, Zolfaghari E, Jubran R, Kim JW. Long‐term outcomes of Group D retinoblastoma eyes during the intravitreal melphalan era. Pediatr Blood Cancer. 2017;64(12):e26696. doi:10.1002/pbc.26696
132. Shields CL, Manjandavida FP, Arepalli S, Kaliki S, Lally SE, Shields JA. Intravitreal melphalan for persistent or recurrent retinoblastoma vitreous seeds: preliminary results. JAMA Ophthalmol. 2014;132(3):319–325. doi:10.1001/jamaophthalmol.2013.7666
133. Ji X, Hua P, Li J, Li J, Zhao J, Zhao P. Intravitreal melphalan for vitreous seeds: initial experience in China. J Ophthalmol. 2016;2016:1–7. doi:10.1155/2016/4387286
134. Suzuki S, Aihara Y, Fujiwara M, Sano S, Kaneko A. Intravitreal injection of melphalan for intraocular retinoblastoma. Jpn J Ophthalmol. 2015;59(3):164–172. doi:10.1007/s10384-015-0378-0
135. Shields CL, Douglass AM, Beggache M, Say EAT, Shields JA. Intravitreous chemotherapy for active vitreous seeding from retinoblastoma. Retina. 2016;36(6):1184–1190. doi:10.1097/IAE.0000000000000903
136. Berry JL, Bechtold M, Shah S, et al. Not all seeds are created equal: seed classification is predictive of outcomes in retinoblastoma. Ophthalmology. 2017;124(12):1817–1825. doi:10.1016/j.ophtha.2017.05.034
137. Rao R, Honavar SG, Sharma V, Reddy VAP. Intravitreal topotecan in the management of refractory and recurrent vitreous seeds in retinoblastoma. Br J Ophthalmol. 2018;102(4):490–495. doi:10.1136/bjophthalmol-2017-310641
138. Abramson DH, Ji X, Francis JH, Catalanotti F, Brodie SE, Habib L. Intravitreal chemotherapy in retinoblastoma: expanded use beyond intravitreal seeds. Br J Ophthalmol. 2019;103(4):488–493. doi:10.1136/bjophthalmol-2018-312037
139. Amin S, Rizvi F, Zia N, Ali A, Hamid A, Kumari B. Outcomes of Group D retinoblastoma with resistant vitreous seeds after integration of intravitreal chemotherapy to the treatment protocol. Cureus. 2020;12:11.
140. Yousef YA, Al Jboor M, Mohammad M, et al. Safety and efficacy of intravitreal chemotherapy (melphalan) to treat vitreous seeds in retinoblastoma. Front Pharmacol. 2021;12. doi:10.3389/fphar.2021.696787