Exosomes in Disease Therapy: Plant-Derived Exosome-Like Nanoparticles Current Status, Challenges, and Future Prospects

Introduction
Exosomes are nanoscale extracellular vesicles (40–150 nm) secreted by bacteria, animals, and plants; they have evolved from being viewed as cellular debris to becoming essential regulators of intercellular communication.1 In the past decade (Figure 1), remarkable progress has been achieved in the exploration of the therapeutic potential of exosomes, especially as delivery vehicles for proteins, lipids, and nucleic acids, which mirror the genetic and proteomic signatures of their parent cells.2–21 The unique biological properties of exosomes, including their superior blood–brain barrier (BBD) penetration compared with synthetic nanoparticles, position them as dual-functional agents for diagnostic biomarker detection and targeted therapy.22 Emerging evidence highlights exosome-mediated CRIPR-Cas9 delivery systems that achieve neuron-specific gene editing with enhanced precision in preclinical models.23 The inherent immune stealth characteristics of exsomes,24,25 combined with their target specificity demonstrated in clinical trials,26,27 establish them as prime candidates for precision therapies, surpassing conventional delivery platforms. From immunoregulation to neural regeneration, exosome applications are revolutionizing therapeutic paradigms owing to their multifunctional biological capacity. Alongside exosomes, a diverse array of nanocarriers—such as gelatin, collagen, and zein-based platforms—have also shown tremendous promise in cancer therapy due to their biocompatibility, ease of surface functionalization, and tunable drug release properties. Recent studies have demonstrated that gelatin-based systems enable environment-responsive delivery, while zein nanoparticles offer effective mucosal adhesion and sustained release. These platforms are now increasingly engineered for tumor targeting, immune modulation, and combination delivery of chemotherapeutics and biologics, as evidenced in recent reports.28–31
|
Figure 1 Timeline of exosome research: From germination to diversified development (1967–2024). |
Building on the therapeutic potential of mammalian exosomes in precision medicine, plant-derived nanovesicles have recently emerged as an analogous delivery platform. Despite their evolutionary divergence from mammalian systems, Plant-Derived Exosome-like Nanoparticles, PLNs have conserved structural characteristics, including the following: 40–150 nm size range, lipid bilayer organization, and cargo sorting mechanisms mediated by endosomal sorting complexes required for transport proteins.1,32 These botanical vectors inherit biocompatibility from their plant origins and exhibit 60–80% lower immunogenicity than mammalian exosomes in trials,33 which can be used to address critical safety concerns for clinical translation. PLN membranes contain phylogenetically conserved HSP70 homologs that enable temperature-regulated drug release and offer spatiotemporal precision unmatched by animal-derived vesicles.34 Despite advances in the understanding of PLN biology, key translational challenges persist between laboratory research and clinical implementation. As green nanotechnology platforms, PLNs circumvent ethical issues related to mammalian cell culture.35
Despite notable progress in elucidating the biological roles and therapeutic potential of PLNs, critical barriers hinder their transition from research tools to clinical therapies. The primary obstacles include challenges in attaining scalable production, ensuring the purity and stability of exosome preparations, and addressing immunogenicity and safety concerns. In addition, regulatory pathways for exosome-based therapies remain unclear and thus present further challenges for clinical translation. PLNs, which show considerable promise owing to their inherent biocompatibility and low immunogenicity, require further pharmacokinetic (PK) and biodistribution studies to validate their safety and efficacy. These challenges underscore the urgent need for robust protocols, standardized quality control measures, and comprehensive regulatory frameworks to fully realize the potential of exosome-based therapies.
This review provides a comprehensive overview of the molecular mechanisms underlying PLN biogenesis and cargo sorting along with the current state of therapeutic applications in oncology, neurology, and immunology. This discussion focuses on recent advances in exosome engineering, including enhanced cargo loading techniques, surface modifications for targeted delivery, and stimulus-responsive release mechanisms. This review critically examines technological and regulatory hurdles, integrates insights from the latest engineering innovations, and outlines a clear roadmap for overcoming the existing challenges. The goal is to accelerate the clinical translation of exosome-based platforms and facilitate their integration into precision medicine and eventual establishment as next-generation tools for disease management.
PLN Biology and Mechanisms of Action
Biogenesis of PLNs
PLNs are increasingly recognized as functionally active extracellular vesicles sharing structural similarities with mammalian exosomes. However, these nanoparticles are generated via unique biosynthetic pathways. Three primary mechanisms are responsible for PLN formation: the multivesicular body (MVB) pathway, the exocyst-positive organelle (EXPO) pathway, and vacuole–plasma membrane fusion, and each contributes distinctively to vesicle formation and cargo loading.36,37 Among the proposed pathways, the MVB pathway is the principal route for PLN biogenesis (Figure 2).38,39 This process is initiated by inward budding of the plasma membrane, which results in the formation of early endosomes. These endosomes mature and interact with the trans-Golgi network, leading to MVB development. Within MVBs, inward invagination of the endosomal membrane generates intraluminal vesicles (ILVs), which serve as carriers for selectively loaded biomolecules, including RNAs, DNAs, lipids, and proteins. Upon fusion of MVBs with the plasma membrane, ILVs are released into the extracellular milieu as PLNs and contribute to intercellular and interorganismal communication.40,41 The MVB pathway, which is well established in mammalian systems, also operates in plants and has been explicitly demonstrated in Arabidopsis thaliana by identifying TET8-positive vesicles.42–45 These plant vesicles are functional analogs of mammalian CD63-positive exosomes and are particularly induced during pathogen attacks, facilitating the secretion of small RNAs and immune regulatory molecules crucial in plant defense responses.46,47
 |
Figure 2 Schematic illustration of PLN biogenesis pathways in plants: MVB-dependent secretion of TET8+ vesicles, EXPO-mediated direct release, and vacuole–plasma membrane fusion under biotic stress. |
The EXPO pathway represents a plant unconventional secretion mechanism that bypasses the classical endoplasmic reticulum–Golgi–endosome system.48 It involves the formation of double-membraned vesicles that directly fuse with the plasma membrane, resulting in the release of their contents into the apoplast. Unlike vesicles derived from the MVB pathway, EXPOs do not associate with canonical endosomal markers, which underscores their independence from endosomal trafficking routes.49 The EXPO pathway plays a critical role in the mediation of the rapid extracellular trafficking of defense-related proteins and signaling molecules in response to abiotic stresses, such as drought, salinity, and oxidative stress.49 These findings mark a notable shift in our comprehension of the functional landscape of plant extracellular vesicle biogenesis, which extends the relevance of EXPO pathway beyond developmental signaling and pathogen response. In Arabidopsis thaliana, stress-induced activation of EXPO vesicle secretion enhances apoplastic reactive oxygen species scavenging and salt tolerance through targeted delivery of antioxidant enzymes and chaperones.50 This outcome supports a novel model in which EXPOs act as first responders to environmental extremes, which rapidly reprograms the extracellular milieu. Furthermore, EXPO-derived plant-derived exosome-like nanoparticles, PLNs have shown an increasing potential as therapeutic nanocarriers. Given their origin in stress-adapted pathways, these vesicles exhibit enhanced stability under physiological conditions and can encapsulate stress-induced metabolites and RNAs with pharmacological relevance.51 They can bypass conventional secretion, which makes them especially valuable for the delivery of hydrophobic or structurally labile therapeutic agents. Recent engineering approaches have explored how the EXPO pathway can be harnessed or mimicked synthetically to design plant-based delivery systems for human applications, including targeted immunotherapy and metabolic disorder modulation.50,51 These advances underscore the EXPO pathway not only as a unique secretion mechanism but also as a biotechnological gateway for stress-responsive bioactive compound delivery.
The third pathway involves direct fusion between vacuoles and the plasma membrane, predominantly under biotic stress.52 This fusion event results in the extracellular release of hydrolytic enzymes and defense-related proteins, which provides immediate local defense mechanisms against pathogens and environmental stressors.47,53,54
Molecular Composition and Bioactive Cargo of PLNs
PLNs have been isolated from a diverse range of plant species, including grapefruit, ginger, and broccoli (Table 1).55–57 These nanoparticles exhibit remarkable biostability and intrinsic biocompatibility, which increases their potential for therapeutic applications. In addition, PLNs are inherently enriched with small regulatory RNAs, secondary metabolites, such as polyphenols and flavonoids, and specific plant defense proteins, which underscores their biological importance in plant physiology and translational potential in biomedical and agricultural applications.5,58
 |
Table 1 Representative Molecular Components of PLNs and Biological Functions |
PLNs are nanovesicles enveloped by lipid bilayers and endowed with diverse cargos that confer distinct bioactivity. These cargos encompass lipids, proteins, nucleic acids, and small-molecule metabolites, each of which plays a pivotal role in intercellular communication, immunomodulation, and disease modulation (Table 1). The functional complexity of PLNs arises from the synergistic effects of their molecular constituents, the composition of which varies depending on the plant source, extraction method, and vesicle sub-population.
Lipids: Structure, Function, and Biological Targeting
Lipids form the structural backbone of PLNs and actively participate in vesicle formation, fusion, and cellular uptake.82 Lipidomic profiling revealed the enrichment of PLNs from diverse plant species with phosphatidic acid (PA), phosphatidylcholine (PC), digalactosyldiacylglycerol, monogalactosyldiacylglycerol, and glycosylinositolphosphoceramides.83–86 Notably, PLNs are devoid of cholesterol, which distinguishes them from mammalian exosomes.86 PA, which is abundant in ginger-derived PLNs (GELNs), promotes the expression of Foxa2 in intestinal epithelial cells and facilitates specific uptake by gut microbiota, such as Lactobacillus rhamnosus, which contributes to gut homeostasis and anti-obesity effects.87 By contrast, PC-rich grapefruit PLNs preferentially accumulate in liver tissue, which enhances hepatic targeting capabilities.88,89 Ceramides in ginseng and Arabidopsis PLNs activate toll-like receptor 4 pathways, which results in the modulation of macrophage polarization and offering antitumor potential.90–92
Proteins: Structural and Functional Mediators
Despite the generally lower protein content of PLNs than that of mammalian exosomes, several cytosolic and membrane-associated proteins, including actin, aquaporins, patellin-3-like proteins (PTL3), clathrin, and heat shock proteins, have been consistently identified.47,93–95 These proteins are implicated in vesicle biogenesis, cargo sorting, and endocytic uptake. Clathrin and PTL3 are highly expressed in citrus-derived PLNs and are associated with vesicle trafficking.1 Garlic-derived vesicles express lectins that specifically bind CD98 receptors on HepG2 cells, which facilitate targeted uptake and anti-inflammatory responses.68 Furthermore, ALIX, a classical mammalian exosome marker, was detected in EVs from germinated kiwi pollen (Actinidia chinensis Planch.), suggesting conserved biogenetic pathways across the kingdoms.96
Nucleic Acids: Cross-Kingdom Regulatory Agents
PLNs encapsulate diverse nucleic acids, including miRNAs, siRNAs, and occasionally DNA.97,98 These RNAs perform regulatory functions in both the plant and mammalian systems. miRNAs from ginger PLNs, such as Mdo-miR7267-3p, target microbial genes to regulate gut microbiota; meanwhile, Osa-miR-530-5p interferes with SARS-CoV-2 replication by blocking ORF1ab translation.88,99 In a survey of 11 plant species, over 400 distinct miRNAs were identified, with some common across species (“frequent” miRNAs) and others species-specific (“rare” miRNAs).5,69,100 These miRNAs are predicted to target mammalian inflammatory and oncogenic pathways, which enables PLNs to act as natural regulators in therapeutic contexts.
Secondary Metabolites: Synergistic Therapeutic Constituents
Beyond macromolecules, PLNs deliver bioactive secondary metabolites, such as 6-gingerol, curcumin, sulforaphane, polyphenols, and ginsenosides (Figure 3).101 These compounds enhance PLNs’ anti-inflammatory, antioxidant, and anticancer properties of PLNs. Broccoli-derived PLNs enriched in sulforaphane activate AMP-activated protein kinase pathways to ameliorate colitis,56 and turmeric PLNs downregulate nuclear factor (NF)-κB signaling to reduce hepatic inflammation.102 Compounds, such as 6-shogaol in ginger PLNs, accumulate in lipid membranes and exhibit dose-dependent therapeutic efficacy against gastrointestinal disorders.77 Similarly, Pueraria lobata PLNs deliver puerarin to promote M2 macrophage polarization and modulate immune responses.103
 |
Figure 3 Continued. |
 |
Figure 3 Continued. |
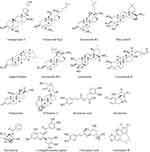 |
Figure 3 Continued. |
 |
Figure 3 Structural Composition and Molecular Cargo of PLNs. |
Technology Platform of PLNs
The advancement of PLNs as therapeutic platforms depends on the development of efficient isolation and purification methods (Figure 4). These procedures are crucial for obtaining high-purity, high-yield, biologically active PLNs for drug delivery, diagnostics, and other clinical applications. With the growing interest in PLNs, existing separation methods need to be adapted and optimized, particularly those that consider the unique properties of plant cells. This section outlines the various technologies employed in PLN isolation, critically assesses their limitations and adaptability to PLNs, and presents future directions for the development of more efficient and scalable technologies (Table 2).
 |
Table 2 Comparison of Production Methods for PLNs, Highlighting Scalability and Purity Challenges |
 |
Figure 4 Isolation and purification methods of PLNs. |
Differential Centrifugation: A Traditional Approach
Differential centrifugation remains the most commonly used method for the isolation of exosomes, including PLNs.65 Through the application using a series of centrifugation steps at different speeds. Initially, low-speed centrifugation removed intact cells and high-speed centrifugation eliminated larger organelles. The final step involves ultracentrifugation to precipitate PLNs from the supernatant. Although this method is simple, scalable, and widely used, its primary limitation lies in time consumption and the risk of structural damage to PLNs due to prolonged centrifugation.123 The addition of a cushion at the bottom of the centrifuge tube can mitigate a portion of the damage to the exosome structure. However, this method requires optimization to achieve a higher purity and gentler processing. This approach remains indispensable, particularly when dealing with large volumes of plant material.
Density Gradient Centrifugation: Enhancing Purity
Density gradient centrifugation is commonly employed.123 This technique uses a gradient of sucrose or other density agents to separate PLNs based on their buoyant density, is commonly employed to enhance the purity of PLNs isolated via differential centrifugation. Typically, a sucrose gradient is created, with PLNs showing enrichment in the 1.13–1.19 g/mL density range. This method is highly effective for isolating pure PLNs, which makes it particularly valuable for applications that require high purity and structural integrity. However, this process is labor intensive and time consuming, which limits its scalability for industrial applications.59 Furthermore, density gradient centrifugation may fail to fully address the problem of contaminating macromolecules or secondary metabolites commonly found in plant materials, which can affect the purity of the final product.
Ultrafiltration: Efficient and Scalable
Ultrafiltration offers a more efficient alternative to traditional methods, particularly when dealing with large sample volumes.104 Through the use of semipermeable membranes with specific molecular weight cutoffs, ultrafiltration separates PLNs based on their size, which allows for their rapid concentration and purification. This method is advantageous for processing large amounts of plant material and is widely applied in research and therapeutic development.61 However, it is insufficient for eliminating small impurities, such as proteins or lipoproteins, which may co-purify with PLNs. Therefore, this method is typically used in combination with other techniques such as differential centrifugation to achieve high purity.
Size Exclusion Chromatography (SEC): Preserving Biological Activity
SEC is particularly beneficial for PLNs because it preserves their biological activity during separation.62 SEC separates particles based on their size as they move through a column filled with porous beads. Larger particles, such as PLNs, elute faster, and smaller contaminants are retained longer. Unlike ultracentrifugation, SEC does not subject PLNs to high mechanical forces, preserves their integrity and biological functionality.72 Furthermore, SEC is scalable, cost-effective, and suitable for the industrial-scale production of PLNs, making it a promising technique for large-scale therapeutic applications. While SEC offers structural preservation, other simpler or cost-efficient strategies, such as precipitation-based methods, also warrant exploration for broader applications.
Polyethylene Glycol (PEG) Precipitation: A Simple and Effective Method
PEG precipitation is a straightforward and cost-effective method for concentrating PLNs.107 By inducing aggregation through PEG, PLNs can be precipitated and isolated through low-speed centrifugation. This method is highly effective for high-yield production, particularly when working with a small sample size. However, PEG precipitation can lead to the aggregation of PLNs with other impurities, which can complicate downstream applications requiring highly purified PLNs.1 To address this issue, researchers often use PEG precipitation in conjunction with other separation techniques to increase the purity of the final product.
Immunoaffinity Separation: Targeted Isolation
Immunoaffinity separation offers a highly specific approach for the isolation of PLNs based on surface markers.110 This method employs antibodies that selectively bind to specific proteins or molecules on the surface of PLNs, which allows for their targeted isolation from complex biological samples. Although this technique is highly effective for mammalian-derived exosomes, the application of immunoaffinity separation for PLNs is currently limited by the lack of well-characterized surface markers. Research on the identification and targeting of specific plant-derived surface proteins is essential to optimize this method for PLN isolation. Immunoaffinity separation can substantially improve the specificity and purity of PLNs for targeted drug delivery and biomarker discovery.
Innovative Technologies and Future Directions
In addition to traditional methods, emerging technologies, such as microfluidics,111 electrophoresis,1 and field-flow fractionation,108 offer new possibilities for the isolation and characterization of PLNs. Microfluidics enables the precise control of fluid flow, which allows for the separation of PLNs with high efficiency and minimal sample loss. Electrophoresis and field-flow fractionation further improve separation specificity by applying electric or flow fields to isolate PLNs based on their size or charge. These innovative technologies are not only more efficient but also display potential for real-time monitoring and on-chip analysis, which are highly desirable for clinical and industrial applications. In the future, integration of multiple separation techniques into a single platform could lead to more efficient, scalable, and cost-effective PLN isolation systems. These advancements are crucial for the widespread application of PLNs in biomedicine, especially in drug delivery, vaccine development, and gene therapy.
Therapeutic Applications of Exosomes
PLNs are prominent vehicles for the advancement of drug delivery,124 gene therapy,125 immunomodulation,126 and cancer treatment (Table 3).127 PLNs, with their unique biogenesis and functional properties, offer unparalleled advantages in precision medicine owing to their capability to carry and deliver a diverse range of bioactive molecules, including nucleic acids, proteins, lipids, and small-molecule drugs. As illustrated in Figure 5, PLNs are involved in various therapeutic domains. The following sections elaborate on their specific applications, beginning with targeted drug delivery.
 |
Table 3 Comparison of Exosomes and PLNs in Major Therapeutic Applications |
 |
Figure 5 PLNs in therapeutic applications. |
Targeted Drug Delivery
Exosomes have garnered increasing recognition for their intrinsic ability to be effective drug delivery systems.137 Their natural biocompatibility, coupled with their capacity to cross physiological barriers, such as the BBB, are ideal candidates for targeted drug delivery.138 The molecular composition of exosomal membranes, which includes cell-specific surface proteins and lipids, enables them to selectively interact with target cells, leading to enhanced cellular uptake and therapeutic efficacy. Moreover, the ability of exosomes to encapsulate hydrophobic and hydrophilic drugs has expanded their application across a broad spectrum of drug types, further supporting their potential as powerful therapeutic vectors. Recent breakthroughs include the successful use of exosomes for the targeted delivery of antitumor agents, which leads to enhanced therapeutic outcomes with reduced systemic toxicity.139
PLNs, which share structural and functional characteristics with mammalian exosomes, represent a promising alternative to traditional drug delivery vehicles.21 These plant-derived nanoparticles offer several key advantages, including nontoxicity, low immunogenicity, and natural bioactive compound encapsulation, which makes them particularly attractive for therapeutic applications.82 PLNs from ginger, a widely studied plant, can encapsulate bioactive molecules, such as curcumin and 6-gingerol, both of which exhibit potent anti-inflammatory and anticancer properties.140,141 The incorporation of these molecules into PLNs enhances bioavailability and targeted delivery to sites of inflammation or tumors. Notably, PLNs cannot deliver small molecules, but they also effectively encapsulate nucleic acids, which enables gene silencing or RNA interference in specific tissues.142 This versatility, combined with their inherent biocompatibility, makes PLNs an ideal platform for personalized medicine and targeted therapies, particularly in conditions where conventional drug delivery methods face limitations. Nonetheless, clinical translation requires careful resolution of key hurdles such as pharmacokinetic variability, immune clearance in human circulation, and dose equivalence between murine models and human patients. Currently, there is no established framework to determine whether effective preclinical doses can be scaled to therapeutic ranges in humans without compromising safety or efficacy.
Gene Therapy
Exosomes have emerged as promising tools for gene delivery, particularly in gene editing technologies such as CRISPR-Cas9.143 Their ability to encapsulate large molecular payloads, such as DNA, RNA, and gene-editing machinery, enables them to function as efficient delivery systems for targeted gene therapy. A notable application is the treatment of genetic diseases, where exosomes loaded with CRISPR-Cas9 complexes can be used to edit specific genes in target cells.144 Exosomes have been used to deliver CRISPR-Cas9 constructs in Alzheimer’s disease models, achieving targeted genome editing with a high efficiency.23 Exosomes, by virtue of their natural composition and cell-specific targeting, can navigate the cellular membrane and deliver gene-editing tools into cells with minimal off-target effects, offering significant advantages over traditional viral vectors and synthetic nanoparticles.
The potential of PLNs in gene therapy is being increasingly recognized, particularly for their ability to carry and deliver genetic material. Given their ability to encapsulate and protect RNA, siRNA, or DNA, PLNs can be used to facilitate gene silencing or gene editing in a variety of contexts.97 While direct evidence for CRISPR-Cas9 delivery via PLNs is currently limited, recent studies have shown that PLNs from sources such as ginger, broccoli, and grapefruit can successfully deliver siRNAs to specific cell types to silence target genes.5,69,73,100,145,146 For example, ginger-derived exosome-like nanoparticles (GELNs) have been used to downregulate pro-inflammatory cytokine expression in inflammatory disease models, demonstrating their capacity to deliver functional nucleic acids.140 Moreover, their excellent biocompatibility, low immunogenicity, and gastrointestinal stability support their promise as next-generation gene delivery vehicles.1 However, in contrast to mammalian exosomes, PLNs have not yet demonstrated equivalent gene editing efficiency for large cargoes such as CRISPR-Cas9 complexes in vivo. This gap is likely due to structural limitations and the current lack of advanced loading and targeting techniques for PLNs. Continued engineering efforts and proof-of-concept studies are needed to explore PLN-mediated CRISPR delivery and optimize their use for precise genome editing. If successful, PLNs may offer a scalable, plant-based alternative to mammalian systems, particularly for oral or mucosal delivery platforms in gene therapy.
Immunotherapy and Inflammatory Disease Treatment
Exosomes play a pivotal role in immune regulation, making them valuable tools for immunotherapy.147 They can modulate immune cell activity by transferring bioactive molecules such as proteins, lipids, and RNAs to recipient cells.
The anti-inflammatory properties of PLNs make them an attractive platform for the treatment of inflammatory diseases.74,103,148 Plant-derived nanoparticles, such as those from grapefruit, can be taken up by intestinal macrophages, which exert well-established anti-inflammatory effects, up-regulate the expression of heme oxygenase-1 and interleukin (IL)-10 in macrophages, and inhibit the secretion of inflammatory cytokines IL-1β and tumor necrosis factor-α.129 Ginger-derived PLNs (GDNPs) have also been shown by multiple research groups to reduce intestinal inflammation and modulate immune responses. For instance, Zhuang reported that GDNPs could protect against DSS-induced colitis by targeting the colon and restoring barrier function.149 Likewise, Teng demonstrated that GDNPs deliver shRNA effectively to intestinal macrophages, suppressing TNF-α and IL-6 expression.88 Turmeric-derived PLNs alleviate symptoms of inflammatory bowel disease and rheumatoid arthritis by reducing systemic inflammation and restoring immune balance.102 Similarly, GELNs have demonstrated efficacy in reducing inflammation in conditions such as osteoarthritis and colitis, rendering them an exciting therapeutic option for inflammatory diseases.77
Cancer Therapy
Exosomes are gaining traction in cancer therapy, particularly because of their ability to deliver chemotherapeutic agents and gene-editing tools to tumor cells.150 PLNs are emerging as effective anticancer agents because of their natural bioactive properties. PLNs derived from broccoli,82 ginger,149 and ginseng90 have shown promising antitumor effects, which are attributed to the presence of compounds, such as sulforaphane, 6-gingerol, and ginsenosides. These compounds exhibit antioxidant, anti-inflammatory, and apoptosis-inducing activities, which collectively contribute to the inhibition of tumor growth and metastasis. In particular, sulforaphane modulates the tumor microenvironment by activating pathways such as AMPK, which leads to enhanced tumor cell death and reduced tumor growth.151 In addition, PLNs derived from ginseng can inhibit tumor progression by modulating immune responses and reducing inflammation.133,152 These findings underscore the potential of PLNs as novel, natural anticancer therapies, either alone or in combination with conventional treatments. Despite these encouraging results, human tumors exhibit higher heterogeneity and complexity than mouse models. Key translational issues, such as intratumoral distribution, endosomal escape, and retention kinetics in solid tumors, remain largely unresolved. Clinical trials are essential to determine whether the antitumor effects of PLNs are replicable in human oncology settings.
Regenerative Medicine
Therefore, exosomes have promising applications in regenerative medicine. These nanocarriers, which can deliver bioactive molecules such as growth factors, cytokines, lipids, and nucleic acids, contribute to stimulating processes such as cell proliferation, migration, and differentiation, which are crucial for tissue repair and regeneration.153,154 However, the real breakthrough in regenerative medicine lies in PLNs, which offer advantages far beyond those of animal-derived exosomes. PLNs, including ginger, turmeric, and ginseng, have significant therapeutic potential owing to their natural biocompatibility, non-toxicity, and ability to encapsulate a wide range of bioactive compounds that promote tissue repair. These nanoparticles not only deliver small molecules such as curcumin, 6-gingerol, and sulforaphane, which are known for their potent anti-inflammatory and regenerative properties, but also encapsulate nucleic acids, facilitating gene silencing or gene editing. The delivery of these compounds through PLNs enhances their bioavailability and enables targeted delivery to damaged tissues, greatly improving the therapeutic outcomes (Figure 6). PLNs derived from ginseng and wheat have demonstrated substantial regenerative capabilities, notably by stimulating endothelial cell migration and promoting angiogenesis, which accelerates wound repair and tissue regeneration.155,156 Furthermore, dandelion-derived PLNs exhibit potent therapeutic potential by effectively neutralizing exotoxins secreted by Staphylococcus aureus.157 In bone regeneration, PLNs have shown potential for the delivery of growth factors and cytokines, improve osteoblast differentiation, and promote bone healing. Moreover, ginseng-derived PLNs have been used to modulate immune responses, reduce inflammation, and inhibit tumor progression, which further enhance their regenerative potential.156 The use of PLNs in regenerative medicine is particularly appealing due to their ability to promote tissue repair without the risk of toxic side effects, which makes them a safer and more effective alternative to traditional therapies.
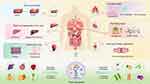 |
Figure 6 Schematic showing how PLNs contribute to tissue repair and regeneration by delivering growth factors and cytokines. |
Challenges in Clinical Translation
PLNs represent an exciting frontier in nanomedicine because they offer inherent biocompatibility, low immunogenicity, and bioactive compound delivery. However, several persistent bottlenecks must be addressed before PLNs can transition from laboratory proof-of-concept to clinical-grade, regulatory-approved therapeutics. These challenges span technical, biological, and regulatory domains, including scalability, standardization, loading efficiency, and the lack of a comprehensive regulatory framework.
Scalability and Production
One of the most critical and persistent barriers to the clinical application of PLNs is the development of scalable, reproducible, and economically sustainable manufacturing platforms that meet the rigorous demands of pharmaceutical production. Traditional techniques used to isolate extracellular vesicles, such as ultracentrifugation,88 SEC,158 and immunoaffinity capture,104 although effective in small-scale research settings, are unsuitable for industrial-scale Good Manufacturing Practice (GMP) production due to their high cost, time consumption, and poor yield reproducibility (Table 2). In ultracentrifugation-based protocols, for example, small deviations in speed, duration, or rotor type can significantly impact vesicle recovery and structural integrity.159 SEC, while providing better separation resolution, scales poorly due to column capacity limitations and often requires trade-offs between throughput and purity.72 Immunoaffinity methods offer specificity but are cost-prohibitive and difficult to standardize for large-volume processing. Compared to synthetic nanoparticles, PLNs are often considered more cost-effective due to their natural abundance and plant-based origin, which reduce raw material expenses. However, this cost advantage can be significantly offset by the complexity and inefficiency of current purification and isolation methods. Synthetic nanoparticles benefit from well-established, scalable, and automated production processes, while PLN production still suffers from low yields, labor-intensive protocols, and batch-to-batch variability. Therefore, when considering total production costs—including downstream processing and quality control—PLNs may not currently offer a significant economic advantage over synthetic alternatives. To address these issues, the field is increasingly exploring emerging technologies such as tangential flow filtration (TFF), microfluidics-based continuous separation, and automated high-throughput isolation platforms. TFF, in particular, is gaining momentum for its compatibility with GMP environments and ability to process large volumes while maintaining vesicle integrity. However, industrial adoption remains limited by technical bottlenecks such as membrane fouling, vesicle retention, and shear stress effects. Moreover, plant cell suspension cultures and controlled-environment agricultural systems (eg, vertical farming) have been proposed for the standardization of PLN production across species and batches.160 These systems offer better control over environmental variables and enable continuous harvesting, which is crucial for batch consistency and regulatory compliance. However, widespread industrialization will require more than technological solutions—it will also necessitate the establishment of process analytical technologies (PAT), validated quality control assays for vesicle identity and purity, and real-time monitoring protocols compatible with GMP standards. Lessons learned from other biologics (eg, vaccines, monoclonal antibodies, enzyme therapies) suggest that early integration of scalable manufacturing design with regulatory compliance pathways is critical to reduce translational delays and downstream reengineering costs.
Purity, Contamination, and Regulatory Issues
Purity and compositional consistency are among the most critical concerns in the clinical translation of PLNs.21 Given the intrinsic biological complexity of plant systems, PLN preparations are often contaminated with cellular debris, lipoproteins, and other non-vesicular particles. Furthermore, their bioactive content and physicochemical properties can vary considerably depending on the plant species, cultivar, growth conditions, and extraction processes.161,162 This lack of formulation uniformity poses a direct threat to clinical reproducibility and safety.
A particularly urgent and unresolved issue is the undefined in vivo pharmacokinetic (PK) profile of PLNs. Despite growing evidence of the systemic bioavailability and cellular uptake of PLNs, their absorption, distribution, metabolism, and excretion characteristics remain largely unexplored. Without clear PK data, dosing regimens and assessment of long-term biodistribution or prediction of off-target effects—factors that are essential for any clinical-grade therapeutic product—become extremely challenging. Moreover, batch-to-batch consistency in PLN production is difficult to achieve under current protocols, especially when crude plant extracts are used as starting materials. This variability can affect not only therapeutic efficacy but also safety and regulatory concerns. Current manufacturing techniques lack validated quality control measures to ensure compositional stability, vesicle-size homogeneity, and functional integrity.
Given these technical limitations, international regulatory standards tailored to PLNs must be established urgently. Although agencies such as the US Food and Drug Administration (FDA) and the European Medicines Agency (EMA), have commenced formulating guidelines for mammalian-derived exosomes, plant-based nanovesicles currently lack clear classification and approval pathways.163 Critical parameters such as minimum identity thresholds, allowable variance in bioactive content, acceptable particle size distribution, and validated analytical methods remain undefined. To move toward clinical-grade applications, comprehensive PK/PD (pharmacokinetics/pharmacodynamics (PD)) profiling, longitudinal safety studies, and global regulatory harmonization are essential. Establishing GMP protocols specific to plant-derived extracellular vesicles, including standardized production pipelines, in-process controls, and post-release specifications, is fundamental for the translation of PLNs into reliable, reproducible, and regulatory-compliant therapeutics.
Stability and Immunogenicity
Another barrier is the physicochemical stability of PLNs during storage and after administration. Factors such as temperature, pH, oxidative degradation, and mechanical stress can affect PLN integrity, which leads to reduced encapsulation efficiency and premature drug release.164 Some therapeutic cargos, such as hydrophobic small molecules or siRNAs, can further destabilize vesicle membranes or aggregates during storage.165 Although plant-derived vesicles generally exhibit lower immunogenicity than mammalian exosomes, their behavior in human hosts is not fully understood. Variables such as the genetic background, gut microbiota composition, and co-administered agents can influence immune activation. Although initial studies have indicated that PLNs are well-tolerated, systematic immunotoxicity testing, allergenicity profiling, and long-term biodistribution tracking are necessary before their clinical use.166
Storage and Delivery Challenges
PLNs are sensitive to temperature, freeze-thaw cycles, and moisture, which makes cold chain logistics a potential bottleneck. Lyophilization and cryoprotectant strategies are being explored to increase shelf life without compromising bioactivity.37,167 Although PLNs demonstrate a certain capability to traverse biological barriers, such as the BBB or intestinal epithelium, their targeting efficiency remains suboptimal. Advances in surface modification, such as the incorporation of ligand-targeting peptides, aptamers, or pH-/enzyme-responsive polymers, can improve biodistribution and enable stimulus-responsive drug release.168 Combining PLNs with hydrogels, microneedles, or oral capsule coatings has emerged as a delivery strategy to increase clinical practicality.
Ethical Issues
The clinical translation of PLN therapies raises ethical concerns, particularly for PLNs. Issues related to long-term safety, potential environmental impacts, and regulatory approval can complicate the widespread adoption of these therapies. Although plant-derived nanoparticles exhibit biocompatibility and low toxicity, their widespread clinical use requires careful ethical considerations, particularly in terms of their manufacturing processes and long-term effects.
PLNs hold great promise for clinical application and offer a wide range of therapeutic possibilities. However, the successful translation of these therapies into clinical practice requires overcoming important challenges, including scalability, purity, stability, and regulatory hurdles. Technological advancements in exosome production, purification, and delivery systems, along with clearer regulatory frameworks, are essential for unlocking the full potential of these therapies. As ongoing research continues to address these challenges, exosome-based and PLN-based therapies are poised to become the cornerstones of personalized medicine and advanced therapeutics.
Technological Innovations and Future Directions
With the continued advancement of PLNs, numerous innovations have propelled their clinical application. However, the focus should be beyond optimistic projections, and the technical limitations, biological uncertainties, and regulatory voids that still constrain their widespread implementation should be critically assessed.
Exosome Engineering: Enhancing Targeting and Delivery
Exosome engineering plays a crucial role in improving the specificity and effectiveness of drug delivery systems (Figure 7).169 By modifying the surfaces of exosomes with specific peptides, antibodies, or aptamers, their targeting capacity can be enhanced, allowing them to deliver therapeutic agents more precisely to target cells or tissues.170,171 Surface modification of exosomes with tumor-targeting antibodies greatly enhances their ability to deliver chemotherapeutic drugs directly to cancer cells, thereby reducing off-target effects and minimizing systemic toxicity.172
 |
Figure 7 Illustration of exosome engineering strategies for enhanced targeting and drug delivery. |
Moreover, advances in synthetic biology have enabled the development of multifunctional exosomes that can simultaneously carry multiple therapeutic agents simultaneously.173 These exosomes can deliver a combination of small molecules, proteins, and nucleic acids, thus offering a more robust therapeutic approach than single-agent therapies. Through the incorporation of stimulus-responsive elements, such as pH-sensitive polymers or light-responsive molecules, exosomes can be engineered to release their cargo in response to specific environmental triggers to provide controlled drug release for better therapeutic outcomes.174 To date, multifunctional exosomes capable of co-delivering small molecules, nucleic acids, and proteins have been investigated for the treatment of complex diseases using synergistic approaches. However, major technical issues still persist. Loading efficiency, cargo stability, and batch-to-batch variability remain largely unresolved. Current cargo-loading strategies—pre-loading during exosome biogenesis and post-loading after secretion—face trade-offs between biological compatibility and scalability. Electroporation and sonication can compromise the membrane integrity, and passive incubation often results in suboptimal payload incorporation. Therefore, a universal, scalable, and cargo-specific loading protocol remains elusive, limiting its clinical standardization. Table 4 summarizes the current exosome engineering strategies and their mechanisms, advantages, limitations, and appropriate uses. A more comprehensive and systematic comparison of these techniques is critical to guide future engineering frameworks.
 |
Table 4 Strategies for Constructing Engineered Exosomes |
Plant-Based Nanoparticles and the Future of Exosome-Like Delivery
PLNs are promising candidates for next-generation drug delivery systems because of their natural abundance, low immunogenicity, and intrinsic pharmacological activity. Compared with mammalian-derived exosomes, PLNs can encapsulate a wider range of plant-derived secondary metabolites, such as flavonoids, alkaloids, and terpenoids, and thus offer therapeutic potential in oncology, inflammation, and neurodegeneration. In addition, plant-derived nanoparticles have demonstrated potential for efficient delivery of small molecules, proteins, and nucleic acids. PLN production is more scalable and cost-effective than exosome production, which makes it an attractive option for large-scale applications. Innovations in plant-based nanoparticle production, such as the use of plant cell cultures or hydroponics, have enabled more efficient and controlled production processes. Furthermore, PLNs are often derived from widely available plant sources, making them accessible for a wide range of therapeutic applications, particularly in resource-limited settings.
Despite these promising features, crucial technical challenges persist. Most critically, PLNs lack well-defined PK and PD profiles, making it difficult to assess their in vivo behavior, therapeutic windows, and long-term biodistribution. Moreover, their formulation consistency remains challenging to control given the biological variability introduced by various plant species, growth conditions, and processing techniques. This inconsistency affects therapeutic efficacy and reproducibility. Current extraction and purification methods are incompatible with GMP standards, which raises concerns regarding the clinical readiness of PLNs. Therefore, future research must prioritize the development of robust, scalable, and reproducible production pipelines coupled with systematic bioanalytical studies to define the safety, efficacy, and mechanistic pathways of PLN action. PLNs can be integrated into clinically relevant exosome-like delivery platforms to address these limitations.
Regulatory Advancements and Clinical Implementation
Clinical translation of PLNs has been hindered by the absence of dedicated regulatory frameworks. Although the FDA and EMA have initiated the development of guidelines for mammalian-derived exosome therapies, PLNs currently exist in a regulatory vacuum.175 A major challenge lies in the intrinsic heterogeneity of PLNs, where variations in plant species, cultivation environments, extraction methods, and post-processing steps can considerably alter nanoparticle composition and bioactivity. For regulatory approval, PLNs must meet stringent criteria for identity, purity, potency, and stability. However, unlike synthetic drugs or mammalian biologics, PLNs often lack a clearly defined mechanism of action and their bioactive payloads are difficult to quantify using existing assays. This dilemma complicates the establishment of batch-to-batch consistency, safety margins, and dosage regimen. Currently, there is no unified consensus on how plant-derived exosome-like nanoparticles should be classified within existing regulatory systems. Under the US FDA framework, PLNs may be evaluated under several categories depending on their intended use and characterization: if designed to treat or prevent disease, they may fall under the botanical drug classification as outlined in the FDA’s Botanical Drug Development Guidance, which applies to complex natural mixtures of plant origin; if they are extensively purified and mechanistically defined, they might alternatively be regulated as biological products; or, if used for general health support without therapeutic claims, they may be treated as dietary supplements. In the European context, the EMA has not yet issued specific guidance for PLNs, but similar to the FDA, they may be considered biological medicinal products if derived from living sources with demonstrated therapeutic activity, potentially falling under the scope of the Committee for Advanced Therapies (CAT). However, in both jurisdictions, no dedicated category currently exists for vesicle-based plant nanotherapeutics, which often leads to fragmented, case-by-case evaluations.
Given this ambiguity, we propose a roadmap to facilitate regulatory approval and translational development of PLNs. First, regulatory agencies should collaborate to establish a working definition of PLNs that clearly distinguishes them from synthetic nanoparticles, crude plant extracts, and conventional biologics. This includes establishing threshold criteria for natural vesicular origin, lipid bilayer structure, and active component characterization. Second, standardization of manufacturing processes—including plant material sourcing, growth conditions, isolation procedures, and purification methods—is essential to ensure consistency and reproducibility. Third, an analytical toolkit for PLN characterization should be developed, incorporating particle size, zeta potential, protein/lipid cargo profiling, and functional assays. Furthermore, tailored safety evaluation protocols—including immunogenicity, biodistribution, and toxicology—must be established specifically for plant-derived nanovesicles. Early regulatory engagement, such as pre-IND (FDA) or scientific advice (EMA), is recommended to clarify classification, clinical trial design, and quality control expectations. Ultimately, international harmonization efforts will be necessary to define acceptable quality attributes, shelf-life, labeling, and clinical indications, thereby supporting the scalable and compliant development of PLNs for therapeutic use.
Personalized Medicine and the Role of Exosome-Based Therapies
PLNs can also have a crucial impact on the development of personalized medicine is another key area.176 Utilizing the natural ability of exosomes and PLNs to carry and deliver a variety of therapeutic agents to specific tissues or organs, these nanoparticles can be engineered to match individual patient profiles. Personalized exosome-based therapies can be developed to deliver gene-editing tools, such as CRISPR-Cas9, directly to target tissues in patients with specific genetic diseases. Similarly, PLNs can be used to deliver personalized cancer therapies targeting specific mutations or tumor types, which can improve the effectiveness of treatment and minimize side effects. Recent advances in genomics and bioinformatics have enabled the identification of specific biomarkers that can be targeted by PLNs.177 The ability to engineer exosomes to carry specific biomolecules that target individual mutations or overexpressed proteins can lead to highly individualized therapeutic strategies.
Future Prospects and Challenges
Although PLNs offer tremendous therapeutic potential, several challenges must be addressed to realize their full capabilities. These challenges include refining the production methods for scalability, improving particle stability, enhancing targeted delivery capabilities, and ensuring long-term safety. Additionally, the lack of clear regulatory pathways and standardized protocols for clinical use remains an important barrier to their widespread adoption. From an ethical and environmental standpoint, the long-term effects of repeated or chronic exposure to PLNs remain poorly understood. Comprehensive studies on their potential bioaccumulation, immunogenicity, and interaction with host cells over extended periods are essential to ensure patient safety. Furthermore, the environmental impact of large-scale PLN production—including agricultural practices, energy consumption, and waste management—must be carefully evaluated to develop sustainable and responsible manufacturing strategies. On a more optimistic note, emerging technologies such as CRISPR-based protein engineering hold great promise for the precise design and functionalization of PLNs. These tools could enable the development of highly customizable delivery platforms with enhanced specificity, reduced immunogenicity, and tailored release profiles. With ongoing technological advancements in exosome engineering, plant-based nanoparticle production, and personalized medicine, these challenges are likely to be progressively overcome.
As the field evolves, PLN-based therapies are poised to play a central role in the future of nanomedicine and offer highly targeted, nontoxic, and efficient therapeutic options for a wide range of diseases. Their unique properties, including biocompatibility, low toxicity, and ability to deliver bioactive molecules, make them key players in the next generation of precision medicine. Continued technological innovation, robust regulatory frameworks, and ethically conscious development will be essential to unlock the full potential of these therapies and transform the landscape of modern medicine.
Conclusion
PLNs are increasingly being recognized as versatile and biocompatible platforms for next-generation therapeutics. Their intrinsic properties, including membrane compatibility, low immunogenicity, and the ability to encapsulate diverse bioactive molecules, enable applications across a broad therapeutic spectrum, from gene editing and immunotherapy to regenerative medicine. PLNs offer distinct advantages, such as oral bioavailability, gastrointestinal stability, and cost-effective scalability, making them especially promising for accessible and sustainable nanomedicine. Recent advances in vesicle engineering have significantly expanded the functional landscapes of exosome-based systems. Surface functionalization, cargo-selective loading, and hybridization with synthetic materials have facilitated precise delivery of miRNAs, siRNAs, CRISPR–Cas9 components, and therapeutic proteins. Clinical trials of several mammalian-derived exosome platforms are ongoing. PLNs have demonstrated unique bioactivities through their enrichment in plant metabolites, proteins, and small RNAs, with therapeutic effects on inflammation, cancer, oxidative stress, and gut immunity. These natural nanocarriers display strong potential not only as standalone therapeutics but also as delivery vehicles synergizing with mRNA vaccines, immune checkpoint inhibitors, and other emerging biologics. However, despite recent advancements in vesicle engineering and biofunctionalization, the field still faces critical scientific bottlenecks that must be addressed to realize their translational potential.
Future Outlook
Moving forward, one of the most pressing challenges is the lack of comprehensive PK and PD characterization of PLNs in human systems. However, the mechanisms governing its absorption, biodistribution, intracellular trafficking, and clearance remain poorly understood. This knowledge gap severely limits the establishment of optimized dosing strategies and safety profiles. Future research should focus on in vivo tracking technologies, such as real-time imaging and labeled tracer systems, to elucidate the behavior of PLNs across biological compartments. Another major limitation is the absence of standardized GMP-compliant production protocols. Current isolation methods lack reproducibility at this scale, which leads to batch-to-batch variability in vesicle composition, purity, and bioactivity. To ensure clinical-grade consistency, researchers should develop automated, scalable production systems, such as plant cell bioreactors or microfluidic isolation platforms, alongside robust quality control assays that can characterize vesicle structures and functional payloads.
Regulatory ambiguity remains a substantial barrier, particularly for PLNs. Although regulatory bodies such as the FDA and EMA have initiated the groundwork for mammalian-derived exosome classification, PLNs currently lack formal regulatory definitions, product categories, and safety testing frameworks. Advancements in clinical approval will necessitate the establishment of internationally harmonized guidelines for plant-derived vesicle therapeutics, including identity thresholds, potency assays, and immunogenicity testing. The integration of exosome-based systems with emerging technologies, such as CRISPR-Cas9 gene editing, mRNA delivery, and tumor microenvironment-targeted immunotherapies, has immense potential. However, such applications will require not only enhanced targeting precision and controlled-release mechanisms, but also a deeper understanding of host–nanoparticle interactions, especially in the context of chronic use, off-target effects, and long-term immunological consequences. Future studies should address these multidimensional challenges by adopting multidisciplinary approaches and accumulating expertise in plant molecular biology, nanotechnology, pharmacology, regulatory science, and systems biology. Additionally, the creation of open-access PLN databases, including vesicle compositions, cargo profiles, and functional readouts, will support global data integration and accelerate innovation.
In summary, PLNs represent a transformative platform for targeted and personalized medicine. However, to move beyond proof-of-concept toward approved therapeutics, the field must address key challenges in standardization, mechanistic clarity, clinical validation, and regulatory alignment. Success in this direction will depend on sustained investment in basic science, engineering innovation, and global collaboration, paving the way for the safe, effective, and equitable use of nanotherapeutics in diverse clinical contexts.
Data Sharing Statement
All data generated or analyzed during this study are included in this published article.
Acknowledgment
This work was financially supported by the National Key R&D Program of China (No. 2023YFC3504402) and Key Project of the National Natural Science Foundation of China (No. 82130113).
Disclosure
The authors declare that they have no known competing financial interests or personal relationships that could influence the work reported in this study.
References
1. Zhou S, Cao Y, Shan F, et al. Analyses of chemical components and their functions in single species plant-derived exosome like vesicle. TrAC Trends Anal Chem. 2023;167:117274. doi:10.1016/j.trac.2023.117274
2. Halperin W, Jensen WA. Ultrastructural changes during growth and embryogenesis in carrot cell cultures. J Ultrastruct Res. 1967;18:428–443. doi:10.1016/S0022-5320(67)80128-X
3. Trams EG, Lauter CJ, Salem NJ, et al. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim Biophys Acta. 1981;645:63–70. doi:10.1016/0005-2736(81)90512-5
4. Pan BT, Johnstone RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell. 1983;33:967–978. doi:10.1016/0092-8674(83)90040-5
5. Vidal M, Sainte-Marie J, Philippot JR, et al. Asymmetric distribution of phospholipids in the membrane of vesicles released during in vitro maturation of Guinea pig reticulocytes: evidence precluding a role for “aminophospholipid translocase”. J Cell Physiol. 1989;140:455–462. doi:10.1002/jcp.1041400308
6. Pinedo ML, Segarra C, Conde RD. Occurrence of two endoproteinases in wheat leaf intercellular washing fluid. Physiol Plant. 1993;88:287–293. doi:10.1111/j.1399-3054.1993.tb05501.x
7. Raposo G, Nijman HW, Stoorvogel W, et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996;183:1161–1172. doi:10.1084/jem.183.3.1161
8. Wolfers J, Lozier A, Raposo G, et al. Tumor-derived exosomes are a source of shared tumor rejection antigens for CTL cross-priming. Nat Med. 2001;7:297–303. doi:10.1038/85438
9. Valadi H, Ekstrom K, Bossios A, et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9:654–659. doi:10.1038/ncb1596
10. Regente M, Corti MG, de la Canal L. Phospholipids are present in extracellular fluids of imbibing sunflower seeds and are modulated by hormonal treatments. J Exp Bot. 2008;59:553–562. doi:10.1093/jxb/erm329
11. Regente M, Corti-Monzon G, Maldonado AM, et al. Vesicular fractions of sunflower apoplastic fluids are associated with potential exosome marker proteins. FEBS Lett. 2009;583:3363–3366. doi:10.1016/j.febslet.2009.09.041
12. Gould SJ. Scientific Program 2012 ISEV meeting Wednesday 18th April. J Extracell Vesicles. 2012;1:18182. doi:10.3402/jev.v1i0.18182
13. Waldenstrom A, Genneback N, Hellman U, et al. Cardiomyocyte microvesicles contain DNA/RNA and convey biological messages to target cells. PLoS One. 2012;7:e34653. doi:10.1371/journal.pone.0034653
14. Supic G. The nobel prize in physiology or medicine 2013. Vojnosanit Pregl. 2013;70:991–992. doi:10.3357/ASEM.3860.2013
15. Lotvall J, Hill AF, Hochberg F, et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: a position statement from the International Society for Extracellular Vesicles. J Extracell Vesicles. 2014;3:26913. doi:10.3402/jev.v3.26913
16. Rutter BD, Rutter KL, Innes RW. Isolation and quantification of plant extracellular vesicles. Biol Protoc. 2017;7:e2533. doi:10.21769/BioProtoc.2533
17. Baldrich P, Rutter BD, Karimi HZ, et al. Plant extracellular vesicles contain diverse small RNA species and are enriched in 10- to 17-Nucleotide “Tiny” RNAs. Plant Cell. 2019;31:315–324. doi:10.1105/tpc.18.00872
18. Liu NJ, Wang N, Bao JJ, et al. Lipidomic analysis reveals the importance of GIPCs in arabidopsis leaf extracellular vesicles. Mol Plant. 2020;13:1523–1532. doi:10.1016/j.molp.2020.07.016
19. Xie Y, Guan Q, Guo J, et al. Hydrogels for exosome delivery in biomedical applications. Gels. 2022;8. doi:10.3390/gels8060328
20. Yin S, Chen A, Ding Y, et al. Recent advances in exosomal RNAs analysis towards diagnostic and therapeutic applications. TrAC Trends Anal Chem. 2023;158:116840. doi:10.1016/j.trac.2022.116840
21. Zhao B, Lin H, Jiang X, et al. Exosome-like nanoparticles derived from fruits, vegetables, and herbs: innovative strategies of therapeutic and drug delivery. Theranostics. 2024;14:4598–4621. doi:10.7150/thno.97096
22. Rohner E, Yang R, Foo KS, et al. Unlocking the promise of mRNA therapeutics. Nat Biotechnol. 2022;40:1586–1600. doi:10.1038/s41587-022-01491-z
23. Han J, Sul JH, Lee J, et al. Engineered exosomes with a photoinducible protein delivery system enable CRISPR-Cas-based epigenome editing in Alzheimer’s disease. Sci Transl Med. 2024;16:i4830. doi:10.1126/scitranslmed.adi4830
24. Cong M, Tan S, Li S, et al. Technology insight: plant-derived vesicles-How far from the clinical biotherapeutics and therapeutic drug carriers? Adv Drug Deliv Rev. 2022;182:114108. doi:10.1016/j.addr.2021.114108
25. Sergazy S, Adekenov S, Khabarov I, et al. Harnessing mammalian- and plant-derived exosomes for drug delivery: a Comparative Review. Int J Mol Sci. 2025;26:4857. doi:10.3390/ijms26104857
26. Group CN. FDA approves direct biologics to proceed with a landmark Phase 3 acute respiratory distress syndrome (ARDS) trial. Chemical News: Official Journal of the Indian Chemical Manufacturers Association. 2022.
27. Yu HR, Huang HC, Chen IL, et al. Exosomes secreted by wharton’s jelly-derived mesenchymal stem cells promote the ability of cell proliferation and migration for keratinocyte. Int J Mol Sci. 2024;25:15. doi:10.3390/ijms25094758
28. Ooi MB, Krueger S, Thomas WJ, et al. Prospective real-time correction for arbitrary head motion using active markers. Magn Reson Med. 2009;62:943–954. doi:10.1002/mrm.22082
29. Madan E, Palma AM, Vudatha V, et al. Ovarian tumor cells gain competitive advantage by actively reducing the cellular fitness of microenvironment cells. Nat Biotechnol. 2024:1–5. doi:10.1038/s41587-024-02453-3
30. Mamidi N, Franco DSF, Orash MA. Advanced disease therapeutics using engineered living drug delivery systems. Nanoscale. 2025;17:7673–7696. doi:10.1039/d4nr05298f
31. Mamidi N, De Silva FF, Vacas AB, et al. Multifaceted hydrogel scaffolds: bridging the gap between biomedical needs and environmental sustainability. Adv Healthc Mater. 2024;13:2401195. doi:10.1002/adhm.202401195
32. Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977. doi:10.1126/science.aau6977
33. Kim J, Li S, Zhang S, et al. Plant-derived exosome-like nanoparticles and their therapeutic activities. Asian J Pharm Sci. 2022;17:53–69. doi:10.1016/j.ajps.2021.05.006
34. Madhan S, Dhar R, Devi A. Plant-derived exosomes: a green approach for cancer drug delivery. J Mater Chem B. 2024;12:2236–2252. doi:10.1039/d3tb02752j
35. Jiang D, Li Z, Liu H, et al. Plant exosome-like nanovesicles derived from sesame leaves as carriers for luteolin delivery: molecular docking, stability and bioactivity. Food Chem. 2024;438:137963. doi:10.1016/j.foodchem.2023.137963
36. Lo KJ, Wang MH, Ho CT, et al. Plant-derived extracellular vesicles: a new revolutionization of modern healthy diets and biomedical applications. J Agric Food Chem. 2024;72:2853–2878. doi:10.1021/acs.jafc.3c06867
37. Pratiwi FW, Thomas RT, Karzarjeddi M, et al. Scalable purification, storage, and release of plant-derived nanovesicles for Local therapy using nanostructured all-cellulose composite membranes. Biomacromolecules. 2024;25:5847–5859. doi:10.1021/acs.biomac.4c00535
38. An Q, van Bel AJE, Hückelhoven R. Do plant cells secrete exosomes derived from multivesicular bodies? Plant Signal Behav. 2007;2:4–7. doi:10.4161/psb.2.1.3596
39. Movahed N, Cabanillas DG, Wan J, et al. Turnip mosaic virus components are released into the extracellular space by vesicles in infected leaves. Plant Physiol. 2019;180:1375–1388. doi:10.1104/pp.19.00381
40. Karamanidou T, Tsouknidas A. Plant-derived extracellular vesicles as therapeutic nanocarriers. Int J Mol Sci. 2021;23:191. doi:10.3390/ijms23010191
41. Li X, Bao H, Wang Z, et al. Biogenesis and function of multivesicular bodies in plant immunity. Front Plant Sci. 2018;9:979. doi:10.3389/fpls.2018.00979
42. Liu C, Yu Y, Fang L, et al. Plant-derived nanoparticles and plant virus nanoparticles: bioactivity, health management, and delivery potential. Crit Rev Food Sci. 2024;64:8875–8891. doi:10.1080/10408398.2023.2204375
43. Willms E, Cabañas C, Mäger I, et al. Extracellular vesicle heterogeneity: subpopulations, isolation techniques, and diverse functions in cancer progression. Front Immunol. 2018;9:738. doi:10.3389/fimmu.2018.00738
44. Hessvik NP, Llorente A. Current knowledge on exosome biogenesis and release. Cell Mol Life Sci. 2018;75:193–208. doi:10.1007/s00018-017-2595-9
45. Thakur A, Ke X, Chen Y, et al. The mini player with diverse functions: extracellular vesicles in cell biology, disease, and therapeutics. Protein Cell. 2022;13:631–654. doi:10.1007/s13238-021-00863-6
46. Boavida LC, Qin P, Broz M, et al. Arabidopsis tetraspanins are confined to discrete expression domains and cell types in reproductive tissues and form homo- and heterodimers when expressed in yeast. Plant Physiol. 2013;163:696–712. doi:10.1104/pp.113.216598
47. Rutter BD, Innes RW. Extracellular vesicles isolated from the leaf apoplast carry stress-response proteins. Plant Physiol. 2017;173:728–741. doi:10.1104/pp.16.01253
48. Wang X, Xin C, Zhou Y, et al. Plant-derived vesicle-like nanoparticles: the next-generation drug delivery nanoplatforms. Pharmaceutics. 2024;16:588. doi:10.3390/pharmaceutics16050588
49. Wang J, Ding Y, Wang J, et al. EXPO, an exocyst-positive organelle distinct from multivesicular endosomes and autophagosomes, mediates cytosol to cell wall exocytosis in Arabidopsis and tobacco cells. Plant Cell. 2010;22:4009–4030. doi:10.1105/tpc.110.080697
50. Cai Q, Qiao L, Wang M, et al. Plants send small RNAs in extracellular vesicles to fungal pathogen to silence virulence genes. Science. 2018;360:1126–1129. doi:10.1126/science.aar4142
51. Zeng YB, Deng X, Shen LS, et al. Advances in plant-derived extracellular vesicles: isolation, composition, and biological functions. Food Funct. 2024;15:11319–11341. doi:10.1039/d4fo04321a
52. He B, Hamby R, Jin H. Plant extracellular vesicles: trojan horses of crosskgingdom warfare. FASEB Bioadv. 2021;3:657–664. doi:10.1096/fba.2021-00040
53. Qiu D, Lange E, Haas TM, et al. Bacterial pathogen infection triggers magic spot nucleotide signaling in arabidopsis thaliana chloroplasts through specific RelA/SpoT homologues. J Am Chem Soc. 2023;145:16081–16089. doi:10.1021/jacs.3c04445
54. Ito Y, Taniguchi K, Kuranaga Y, et al. Uptake of microRNAs from exosome-like nanovesicles of edible plant juice by rat enterocytes. Int J Mol Sci. 2021;22:3749. doi:10.3390/ijms22073749
55. Raimondo S, Naselli F, Fontana S, et al. Citrus limon-derived nanovesicles inhibit cancer cell proliferation and suppress CML xenograft growth by inducing TRAIL-mediated cell death. Oncotarget. 2015;6:19514–19527. doi:10.18632/oncotarget.4004
56. Deng Z, Rong Y, Teng Y, et al. Broccoli-derived nanoparticle inhibits mouse colitis by activating dendritic cell AMP-activated protein kinase. Mol Ther. 2017;25:1641–1654. doi:10.1016/j.ymthe.2017.01.025
57. Mu J, Zhuang X, Wang Q, et al. Interspecies communication between plant and mouse gut host cells through edible plant derived exosome-like nanoparticles. Mol Nutr Food Res. 2014;58:1561–1573. doi:10.1002/mnfr.201300729
58. Simpson RJ, Lim JW, Moritz RL, et al. Exosomes: proteomic insights and diagnostic potential. Expert Rev Proteomic. 2009;6:267–283. doi:10.1586/epr.09.17
59. Cho E, Choi S, Kim H, et al. Panax ginseng-derived extracellular vesicles facilitate anti-senescence effects in human skin cells: an eco-friendly and sustainable way to use ginseng substances. Cells. 2021;10:486. doi:10.3390/cells10030486
60. Gao WJ, Hou M, Cheng XX, et al. Mechanism of astragali radix vesicle-like nanoparticles for reducing blood glucose in db/db diabetic mice by regulating gut microbiota. Chin J Exp Tradit Med Formulae. 2021;27:111–118.
61. Berger E, Colosetti P, Jalabert A, et al. Use of nanovesicles from Orange juice to reverse diet-induced gut modifications in diet-induced obese mice. Mol Ther Methods Clin Dev. 2020;18:880–892. doi:10.1016/j.omtm.2020.08.009
62. Kim DK, Rhee WJ. Antioxidative effects of carrot-derived nanovesicles in cardiomyoblast and neuroblastoma cells. Pharmaceutics. 2021;13:1203. doi:10.3390/pharmaceutics13081203
63. Zu M, Xie D, Canup BSB, et al. ‘Green’nanotherapeutics from tea leaves for orally targeted prevention and alleviation of colon diseases. Biomaterials. 2021;279:121178. doi:10.1016/j.biomaterials.2021.121178
64. Ly NP, Han HS, Kim M, et al. Plant-derived nanovesicles: current understanding and applications for cancer therapy. Bioact Mater. 2023;22:365–383. doi:10.1016/j.bioactmat.2022.10.005
65. Lu X, Han Q, Chen J, et al. Celery (Apium graveolens L.) exosome-like nanovesicles as a new-generation chemotherapy drug delivery platform against tumor proliferation. J Agric Food Chem. 2023;71:8413–8424. doi:10.1021/acs.jafc.2c07760
66. Sriwastva MK, Deng ZB, Wang B, et al. Exosome-like nanoparticles from mulberry bark prevent DSS-induced colitis via the AhR/COPS8 pathway. EMBO Rep. 2022;23:e53365. doi:10.15252/embr.202153365
67. Kim MK, Choi YC, Cho SH, et al. The Antioxidant effect of small extracellular vesicles derived from aloe vera peels for wound healing. Tissue Eng Regen Med. 2021;18:561–571. doi:10.1007/s13770-021-00367-8
68. Song H, Canup B, Ngo VL, et al. Internalization of garlic-derived nanovesicles on liver cells is triggered by interaction with CD98. Acs Omega. 2020;5:23118–23128. doi:10.1021/acsomega.0c02893
69. Xiao J, Feng S, Wang X, et al. Identification of exosome-like nanoparticle-derived microRNAs from 11 edible fruits and vegetables. PeerJ. 2018;6:e5186. doi:10.1186/s12951-021-00995-1
70. Zhuang X, Teng Y, Samykutty A, et al. Grapefruit-derived nanovectors delivering therapeutic miR17 through an intranasal route inhibit brain tumor progression. Mol Ther. 2016;24:96–105. doi:10.1038/mt.2015.188
71. Du J, Liang Z, Xu J, et al. Plant-derived phosphocholine facilitates cellular uptake of anti-pulmonary fibrotic HJT-sRNA-m7. Sci China Life Sci. 2019;62:309–320. doi:10.1007/s11427-017-9026-7
72. You JY, Kang SJ, Rhee WJ. Isolation of cabbage exosome-like nanovesicles and investigation of their biological activities in human cells. Bioact Mater. 2021;6:4321–4332. doi:10.1016/j.bioactmat.2021.04.023
73. Yang M, Luo Q, Chen X, et al. Bitter melon derived extracellular vesicles enhance the therapeutic effects and reduce the drug resistance of 5-fluorouracil on oral squamous cell carcinoma. J Nanobiotechnology. 2021;19:259. doi:10.1186/s12951-021-00995-1
74. Ishida T, Kawada K, Jobu K, et al. Exosome-like nanoparticles derived from Allium tuberosum prevent neuroinflammation in microglia-like cells. J Pharm Pharmacol. 2023;75:1322–1331. doi:10.1093/jpp/rgad062
75. Perut F, Roncuzzi L, Avnet S, et al. Strawberry-derived exosome-like nanoparticles prevent oxidative stress in human mesenchymal stromal cells. Biomolecules. 2021;11:87. doi:10.3390/biom11010087
76. Chen X, Liu B, Li X, et al. Identification of anti-inflammatory vesicle-like nanoparticles in honey. J Extracell Vesicles. 2021;10:e12069. doi:10.1002/jev2.12069
77. Zhang M, Viennois E, Prasad M, et al. Edible ginger-derived nanoparticles: a novel therapeutic approach for the prevention and treatment of inflammatory bowel disease and colitis-associated cancer. Biomaterials. 2016;101:321–340. doi:10.1016/j.biomaterials.2016.06.018
78. Teng Y, Luo C, Qiu X, et al. Plant-nanoparticles enhance anti-PD-L1 efficacy by shaping human commensal microbiota metabolites. Nat Commun. 2025;16:1295. doi:10.1038/s41467-025-56498-2
79. Wei Y, Li M, Cui S, et al. Shikonin inhibits the proliferation of human breast cancer cells by reducing tumor-derived exosomes. Molecules. 2016;21:777. doi:10.3390/molecules21060777
80. Lu M, Shih Y, Chien TC, et al. α-Solanine inhibits human melanoma cell migration and invasion by reducing matrix metalloproteinase-2/9 activities. Biol Pharm Bull. 2010;33:1685–1691. doi:10.1248/bpb.33.1685
81. Chen T, Ma B, Lu S, et al. Cucumber-derived nanovesicles containing cucurbitacin B for non-small cell lung cancer therapy. Int J Nanomed. 2022;17:3583–3599. doi:10.2147/IJN.S362244
82. Cao M, Diao N, Cai X, et al. Plant exosome nanovesicles (PENs): green delivery platforms. Mater Horiz. 2023;1:3879–3894. doi:10.1039/d3mh01030a
83. Fang Z, Liu K. Plant-derived extracellular vesicles as oral drug delivery carriers. J Control Release. 2022;350:389–400. doi:10.1016/j.jconrel.2022.08.046
84. Chen YF, Luh F, Ho YS, et al. Exosomes: a review of biologic function, diagnostic and targeted therapy applications, and clinical trials. J Biomed Sci. 2024;31:67. doi:10.1186/s12929-024-01055-0
85. Minciacchi VR, Freeman MR, Di Vizio D. Extracellular vesicles in cancer: exosomes, microvesicles and the emerging role of large oncosomes. Semin Cell Dev Biol. 2015;40:41–51. doi:10.1016/j.semcdb.2015.02.010
86. Dad HA, Gu TW, Zhu AQ, et al. Plant exosome-like nanovesicles: emerging therapeutics and drug delivery nanoplatforms. Mol Ther. 2021;29:13–31. doi:10.1016/j.ymthe.2020.11.030
87. Kumar A, Sundaram K, Teng Y, et al. Ginger nanoparticles mediated induction of Foxa2 prevents high-fat diet-induced insulin resistance. Theranostics. 2022;12:1388–1403. doi:10.7150/thno.62514
88. Teng Y, Ren Y, Sayed M, et al. Plant-derived exosomal microRNAs shape the gut microbiota. Cell Host Microbe. 2018;24:637–652. doi:10.1016/j.chom.2018.10.001
89. Ju S, Mu J, Dokland T, et al. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Mol Ther. 2013;21:1345–1357. doi:10.1038/mt.2013.64
90. Cao M, Yan H, Han X, et al. Ginseng-derived nanoparticles alter macrophage polarization to inhibit melanoma growth. J Immunother Cancer. 2019;7:326. doi:10.1186/s40425-019-0817-4
91. Fischer H, Ellstrom P, Ekstrom K, et al. Ceramide as a TLR4 agonist; a putative signalling intermediate between sphingolipid receptors for microbial ligands and TLR4. Cell Microbiol. 2007;9:1239–1251. doi:10.1111/j.1462-5822.2006.00867.x
92. Bai AP, Guo Y. Ceramide is a potential activator of immune responses against tumors. Gastroenterology. 2018;155:579–580. doi:10.1053/j.gastro.2018.04.037
93. Zhang M, Viennois E, Xu C, et al. Plant derived edible nanoparticles as a new therapeutic approach against diseases. Tissue Barriers. 2016;4:e1134415.
94. Pocsfalvi G, Turiak L, Ambrosone A, et al. Protein biocargo of citrus fruit-derived vesicles reveals heterogeneous transport and extracellular vesicle populations. J Plant Physiol. 2018;229:111–121. doi:10.1016/j.jplph.2018.07.006
95. Garaeva L, Kamyshinsky R, Kil Y, et al. Delivery of functional exogenous proteins by plant-derived vesicles to human cells in vitro. Sci Rep. 2021;11:6489. doi:10.1038/s41598-021-85833-y
96. Suanno C, Tonoli E, Fornari E, et al. Small extracellular vesicles released from germinated kiwi pollen (pollensomes) present characteristics similar to mammalian exosomes and carry a plant homolog of ALIX. Front Plant Sci. 2023;14:1090026. doi:10.3389/fpls.2023.1090026
97. Qin X, Wang X, Xu K, et al. Digestion of plant dietary miRNAs starts in the mouth under the protection of coingested food components and plant-derived exosome-like nanoparticles. J Agric Food Chem. 2022;70:4316–4327. doi:10.1021/acs.jafc.1c07730
98. Li D, Yao X, Yue J, et al. Advances in bioactivity of microRNAs of plant-derived exosome-like nanoparticles and milk-derived extracellular vesicles. J Agric Food Chem. 2022;70:6285–6299. doi:10.1021/acs.jafc.2c00631
99. Teng Y, Xu F, Zhang X, et al. Plant-derived exosomal microRNAs inhibit lung inflammation induced by exosomes SARS-CoV-2 Nsp12. Mol Ther. 2021;29:2424–2440. doi:10.1016/j.ymthe.2021.05.005
100. Kantarcioglu M, Yildirim G, Akpinar OP, et al. Coffee-derived exosome-like nanoparticles: are they the secret heroes?. Turk J Gastroenterol. 2023;34:161–169. doi:10.5152/tjg.2022.21895
101. Wang K, Chen Q, Shao Y, et al. Anticancer activities of TCM and their active components against tumor metastasis. Biomed Pharmacother. 2021;133:111044. doi:10.1016/j.biopha.2020.111044
102. Liu C, Yan X, Zhang Y, et al. Oral administration of turmeric-derived exosome-like nanovesicles with anti-inflammatory and pro-resolving bioactions for murine colitis therapy. J Nanobiotechnology. 2022;20:206. doi:10.1186/s12951-022-01421-w
103. Wu J, Ma X, Lu Y, et al. Edible pueraria lobata-derived exosomes promote M2 macrophage polarization. Molecules. 2022;27:8184. doi:10.3390/molecules27238184
104. Mukerjee N, Bhattacharya A, Maitra S, et al. Exosome isolation and characterization for advanced diagnostic and therapeutic applications. Mater Today Bio. 2025;31:101613. doi:10.1016/j.mtbio.2025.101613
105. Koshy L, Madhuma M, Vyshak Y, et al. Peripheral blood mononuclear cells isolation from whole blood of heart failure patients using density-gradient centrifugation. Heart Failure J India. 2025;2:128–140. doi:10.4103/HFJI.HFJI_26_24
106. Li P, Kaslan M, Lee SH, et al. Progress in exosome isolation techniques. Theranostics. 2017;7:789–804. doi:10.7150/thno.18133
107. Wang Y, Teraoka I, Hansen FY, et al. A theoretical study of the separation principle in size exclusion chromatography. Macromolecules. 2010;43:1651–1659. doi:10.1021/ma902377g
108. Yu HJ, Moon MH. Direct lipid analysis of exosomes in serum by online miniaturized asymmetrical flow field-flow fractionation and electrospray ionization-mass spectrometry: application to extrahepatic cholangiocarcinoma. J Chromatogr A. 2025;1746:465778. doi:10.1016/j.chroma.2025.465778
109. Konoshenko MY, Lekchnov EA, Vlassov AV, et al. Isolation of extracellular vesicles: general methodologies and latest trends. Biomed Res Int. 2018;2018:8545347. doi:10.1155/2018/8545347
110. Zarovni N, Corrado A, Guazzi P, et al. Integrated isolation and quantitative analysis of exosome shuttled proteins and nucleic acids using immunocapture approaches. Methods. 2015;87:46–58. doi:10.1016/j.ymeth.2015.05.028
111. Chen J, Li P, Zhang T, et al. Review on strategies and technologies for exosome isolation and purification. Front Bioeng Biotechnol. 2021;9:811971. doi:10.3389/fbioe.2021.811971
112. Jackson KK, Mata C, Marcus RK. A rapid capillary-channeled polymer (C-CP) fiber spin-down tip approach for the isolation of plant-derived extracellular vesicles (PDEVs) from 20 common fruit and vegetable sources. Talanta. 2023;252:123779. doi:10.1016/j.talanta.2022.123779
113. Chen H, Bai Z, Tao S, et al. Optimization of enzyme-assisted microwave extraction, structural characterization, antioxidant activity and in vitro protective effect against H2O2-induced damage in HepG2 cells of polysaccharides from roots of rubus crataegifolius bunge. Int J Biol Macromol. 2024;276:133969. doi:10.1016/j.ijbiomac.2024.133969
114. Busatto S, Vilanilam G, Ticer T, et al. Tangential flow filtration for highly efficient concentration of extracellular vesicles from large volumes of fluid. Cells. 2018;7:273. doi:10.3390/cells7120273
115. Heinemann ML, Ilmer M, Silva LP, et al. Benchtop isolation and characterization of functional exosomes by sequential filtration. J Chromatogr A. 2014;1371:125–135. doi:10.1016/j.chroma.2014.10.026
116. Ghosh A, Davey M, Chute IC, et al. Rapid isolation of extracellular vesicles from cell culture and biological fluids using a synthetic peptide with specific affinity for heat shock proteins. PLoS One. 2014;9:e110443. doi:10.1371/journal.pone.0110443
117. Wang S, Zhang L, Wan S, et al. Aptasensor with expanded nucleotide using DNA nanotetrahedra for electrochemical detection of cancerous exosomes. Acs Nano. 2017;11:3943–3949. doi:10.1021/acsnano.7b00373
118. Wan Y, Cheng G, Liu X, et al. Rapid magnetic isolation of extracellular vesicles via lipid-based nanoprobes. Nat Biomed Eng. 2017;1:0058. doi:10.1038/s41551-017-0058
119. Kosanovic M, Milutinovic B, Goc S, et al. Ion-exchange chromatography purification of extracellular vesicles. Biotechniques. 2017;63:65–71. doi:10.2144/000114575
120. Corso G, Mager I, Lee Y, et al. Reproducible and scalable purification of extracellular vesicles using combined bind-elute and size exclusion chromatography. Sci Rep. 2017;7:11561. doi:10.1038/s41598-017-10646-x
121. Morales-Kastresana A, Musich TA, Welsh JA, et al. High-fidelity detection and sorting of nanoscale vesicles in viral disease and cancer. J Extracell Vesicles. 2019;8:1597603. doi:10.1080/20013078.2019.1597603
122. Wunsch BH, Smith JT, Gifford SM, et al. Nanoscale lateral displacement arrays for the separation of exosomes and colloids down to 20 nm. Nat Nanotechnol. 2016;11:936–940. doi:10.1038/nnano.2016.134
123. Ren Y, He M, Zeng Q, et al. Efficacy and mechanism evaluation, and medicinal development prospects of plant-derived vesicles in therapeutics of inflammatory bowel disease. Chin Herb Med. 2024;55:8208–8221.
124. Fernandes M, Lopes I, Magalhaes L, et al. Novel concept of exosome-like liposomes for the treatment of Alzheimer’s disease. J Control Release. 2021;336:130–143. doi:10.1016/j.jconrel.2021.06.018
125. Liu S, Chen L, Guo M, et al. Targeted delivery of engineered RVG-BDNF-exosomes: a novel neurobiological approach for ameliorating depression and regulating neurogenesis. Research. 2024;7:0402. doi:10.34133/research.0402
126. Kim WY, Won M, Koo S, et al. Mitochondrial H2Sn-mediated anti-inflammatory theranostics. Nanomicro Lett. 2021;13:168. doi:10.1007/s40820-021-00689-1
127. Li Y, Buckhaults P, Li S, et al. Temporal efficacy of a sulforaphane-based broccoli sprout diet in prevention of breast cancer through modulation of epigenetic mechanisms. Cancer Prev Res. 2018;11:451–464. doi:10.1158/1940-6207.CAPR-17-0423
128. Orefice NS, Di Raimo R, Mizzoni D, Logozzi M, Fais S. Purposing plant-derived exosomes-like nanovesicles for drug delivery: patents and literature review. Expert Opin Ther Pat. 2023;33:89–100. doi:10.1080/13543776.2023.2195093
129. Wang B, Zhuang X, Deng ZB, et al. Targeted drug delivery to intestinal macrophages by bioactive nanovesicles released from grapefruit. Mol Ther. 2014;22:522–534. doi:10.1038/mt.2013.190
130. Wang Q, Zhuang X, Mu J, et al. Delivery of therapeutic agents by nanoparticles made of grapefruit-derived lipids. Nat Commun. 2013;4:1867. doi:10.1038/ncomms2886
131. Shao J, Zaro J, Shen Y. Advances in exosome-based drug delivery and tumor targeting: from tissue distribution to intracellular fate. Int J Nanomed. 2020;15:9355–9371. doi:10.2147/IJN.S281890
132. Li Z, Wang H, Yin H, et al. Arrowtail RNA for ligand display on ginger exosome-like nanovesicles to systemic deliver siRNA for cancer suppression. Sci Rep. 2018;8:14644. doi:10.1038/s41598-018-32953-7
133. Han X, Wei Q, Lv Y, et al. Ginseng-derived nanoparticles potentiate immune checkpoint antibody efficacy by reprogramming the cold tumor microenvironment. Mol Ther. 2022;30:327–340. doi:10.1016/j.ymthe.2021.08.028
134. Seo K, Yoo JH, Kim J, et al. Ginseng-derived exosome-like nanovesicles extracted by sucrose gradient ultracentrifugation to inhibit osteoclast differentiation. Nanoscale. 2023;15:5798–5808. doi:10.1039/d2nr07018a
135. Chen Q, Li Q, Liang Y, et al. Natural exosome-like nanovesicles from edible tea flowers suppress metastatic breast cancer via ROS generation and microbiota modulation. Acta Pharm Sin B. 2022;12:907–923. doi:10.1016/j.apsb.2021.08.016
136. Zhang M, Xiao B, Wang H, et al. Edible ginger-derived nano-lipids loaded with doxorubicin as a novel drug-delivery approach for colon cancer therapy. Mol Ther. 2016;24:1783–1796. doi:10.1038/mt.2016.159
137. Tang TT, Wang B, Wu M, et al. Extracellular vesicle-encapsulated IL-10 as novel nanotherapeutics against ischemic AKI. Sci Adv. 2020;6:z748. doi:10.1126/sciadv.aaz0748
138. Liang X, Fa W, Wang N, et al. Exosomal miR-532-5p induced by long-term exercise rescues blood-brain barrier function in 5XFAD mice via downregulation of EPHA4. Aging Cell. 2023;22:e13748. doi:10.1111/acel.13748
139. Dai J, Su Y, Zhong S, et al. Exosomes: key players in cancer and potential therapeutic strategy. Signal Transduct Target Ther. 2020;5:145. doi:10.1038/s41392-020-00261-0
140. Gao C, Zhou Y, Chen Z, et al. Turmeric-derived nanovesicles as novel nanobiologics for targeted therapy of ulcerative colitis. Theranostics. 2022;12:5596–5614. doi:10.7150/thno.73650
141. Jia G, Han Y, An Y, et al. NRP-1 targeted and cargo-loaded exosomes facilitate simultaneous imaging and therapy of glioma in vitro and in vivo. Biomaterials. 2018;178:302–316. doi:10.1016/j.biomaterials.2018.06.029
142. Cai Q, He B, Wang S, et al. Message in a bubble: shuttling small RNAs and proteins between cells and interacting organisms using extracellular vesicles. Annu Rev Plant Biol. 2021;72:497–524. doi:10.1146/annurev-arplant-081720-010616
143. Wan T, Zhong J, Pan Q, et al. Exosome-mediated delivery of cas9 ribonucleoprotein complexes for tissue-specific gene therapy of liver diseases. Sci Adv. 2022;8:9435. doi:10.1126/sciadv.abp9435
144. Chen R, Huang H, Liu H, et al. Friend or foe? Evidence indicates endogenous exosomes can deliver functional gRNA and Cas9 protein. Small. 2019;15:e1902686. doi:10.1002/smll.201902686
145. Potesta M, Roglia V, Fanelli M, et al. Effect of microvesicles from moringa oleifera containing miRNA on proliferation and apoptosis in tumor cell lines. Cell Death Discov. 2020;6:43. doi:10.1038/s41420-020-0271-6
146. Wang X, Wu B, Sun G, et al. Selenium biofortification enhanced miR167a expression in broccoli extracellular vesicles inducing apoptosis in human pancreatic cancer cells by targeting IRS1. Int J Nanomed. 2023;18:2431–2446. doi:10.2147/IJN.S394133
147. Shen DD, Pang JR, Bi YP, et al. LSD1 deletion decreases exosomal PD-L1 and restores T-cell response in gastric cancer. Mol Cancer. 2022;21:75. doi:10.1186/s12943-022-01557-1
148. Bian Y, Li W, Jiang X, et al. Garlic-derived exosomes carrying miR-396e shapes macrophage metabolic reprograming to mitigate the inflammatory response in obese adipose tissue. J Nutr Biochem. 2023;113:109249. doi:10.1016/j.jnutbio.2022.109249
149. Zhuang X, Deng ZB, Mu J, et al. Ginger-derived nanoparticles protect against alcohol-induced liver damage. J Extracell Vesicles. 2015;4:28713. doi:10.3402/jev.v4.28713
150. Wang X, Huang J, Chen W, et al. The updated role of exosomal proteins in the diagnosis, prognosis, and treatment of cancer. Exp Mol Med. 2022;54:1390–1400. doi:10.1038/s12276-022-00855-4
151. Singh A, Sharma B, Deswal R. Green silver nanoparticles from novel brassicaceae cultivars with enhanced antimicrobial potential than earlier reported brassicaceae members. J Trace Elem Med Biol. 2018;47:1–11. doi:10.1016/j.jtemb.2018.01.001
152. Lv Y, Li M, Weng L, et al. Ginseng-derived nanoparticles reprogram macrophages to regulate arginase-1 release for ameliorating T cell exhaustion in tumor microenvironment. J Exp Clin Cancer Res. 2023;42:322. doi:10.1186/s13046-023-02888-7
153. Wang K, He Q, Yang M, et al. Glycoengineered extracellular vesicles released from antibacterial hydrogel facilitate diabetic wound healing by promoting angiogenesis. J Extracell Vesicles. 2024;13:e70013. doi:10.1002/jev2.70013
154. Kee LT, Ng CY, Al-Masawa ME, et al. Extracellular vesicles in facial aesthetics: a review. Int J Mol Sci. 2022;23:6742. doi:10.3390/ijms23126742
155. Sahin F, Kocak P, Gunes MY, et al. In vitro wound healing activity of wheat-derived nanovesicles. Appl Biochem Biotechnol. 2019;188:381–394. doi:10.1007/s12010-018-2913-1
156. Yang S, Lu S, Ren L, et al. Ginseng-derived nanoparticles induce skin cell proliferation and promote wound healing. J Ginseng Res. 2023;47:133–143. doi:10.1016/j.jgr.2022.07.005
157. Tan S, Liu Z, Cong M, et al. Dandelion-derived vesicles-laden hydrogel dressings capable of neutralizing Staphylococcus aureus exotoxins for the care of invasive wounds. J Control Release. 2024;368:355–371. doi:10.1016/j.jconrel.2024.02.045
158. Chin AR, Fong MY, Somlo G, et al. Cross-kingdom inhibition of breast cancer growth by plant miR159. Cell Res. 2016;26:217–228. doi:10.1038/cr.2016.13
159. Zhao Q, Wang T, Wang H, et al. Consensus statement on research and application of Chinese herbal medicine derived extracellular vesicles-like particles (2023 edition). Chin Herb Med. 2024;16:3–12.
160. Shkryl Y, Tsydeneshieva Z, Menchinskaya E, et al. Exosome-like nanoparticles, high in trans-delta-viniferin derivatives, produced from grape cell cultures: preparation, characterization, and anticancer properties. Biomedicines. 2024;12:2142. doi:10.3390/biomedicines12092142
161. Zhu H, Chang M, Wang Q, et al. Identifying the potential of miRNAs in houttuynia cordata-derived exosome-like nanoparticles against respiratory RNA viruses. Int J Nanomed. 2023;18:5983–6000. doi:10.2147/IJN.S425173
162. Zhao Z, Yu S, Li M, et al. Isolation of exosome-like nanoparticles and analysis of microRNAs derived from coconut water based on small RNA high-throughput sequencing. J Agric Food Chem. 2018;66:2749–2757. doi:10.1021/acs.jafc.7b05614
163. Tan A, De La Pena H, Seifalian AM. The application of exosomes as a nanoscale cancer vaccine. Int J Nanomed. 2010;5:889–900. doi:10.2147/IJN.S13402
164. Li SD, Hou X, Qi HZ, et al. Exosomes: provide naturally occurring endogenous nanocarriers for effective drug delivery strategies. Prog Chem. 2016;28:353–362.
165. Bang S, Park B, Park JC, et al. Exosome-inspired lipid nanoparticles for enhanced tissue penetration. ACS Nano. 2025;19:8882–8894. doi:10.1021/acsnano.4c16629
166. Li C, Ni YQ, Xu H, et al. Roles and mechanisms of exosomal non-coding RNAs in human health and diseases. Signal Transduct Target Ther. 2021;6:383. doi:10.1038/s41392-021-00779-x
167. Karn V, Ahmed S, Tsai LW, et al. Extracellular vesicle-based therapy for COVID-19: promises, challenges and future prospects. Biomedicines. 2021;9:1373. doi:10.3390/biomedicines9101373
168. Yang YY, Ma SN, Chen J, et al. Research progress on exosomes and their engineering in ischemic stroke. J Apoplexy Nerv Dis. 2023;40:667–672.
169. Cui J, Zhang Y, Zhang NK, et al. Research progress of engineered exosomes in drug delivery. Prog Pharm Sci. 2024;48:633–640.
170. Segel M, Lash B, Song J, et al. Mammalian retrovirus-like protein PEG10 packages its own mRNA and can be pseudotyped for mRNA delivery. Science. 2021;373:882–889. doi:10.1126/science.abg6155
171. Reshke R, Taylor JA, Savard A, et al. Reduction of the therapeutic dose of silencing RNA by packaging it in extracellular vesicles via a pre-microRNA backbone. Nat Biomed Eng. 2020;4:52–68. doi:10.1038/s41551-019-0502-4
172. Garofalo M, Villa A, Crescenti D, et al. Heterologous and cross-species tropism of cancer-derived extracellular vesicles. Theranostics. 2019;9:5681–5693. doi:10.7150/thno.34824
173. Guo J, Ding QH, Liu Z, et al. Biological characteristics and immunoregulation of exosomes derived from mesenchymal stem cells. Chin J Tissue Eng Res. 2022;26:1093–1101.
174. Tan F, Li X, Wang Z, et al. Clinical applications of stem cell-derived exosomes. Signal Transduct Target Ther. 2024;9:17. doi:10.1038/s41392-023-01704-0
175. Ocansey DKW, Xinwei XU, Zhang LU, et al. Mesenchymal stem cell-derived exosome: the likely game-changer in stem cell research. Biocell. 2022;46:1169–1172. doi:10.32604/biocell.2022.018470
176. Srivastava A, Babu A, Filant J, et al. Exploitation of exosomes as nanocarriers for gene-, chemo-, and immune-therapy of cancer. J Biomed Nanotechnol. 2016;12:1159–1173. doi:10.1166/jbn.2016.2205
177. Chen G, Huang AC, Zhang W, et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature. 2018;560:382–386. doi:10.1038/s41586-018-0392-8