Nanoporous Materials in Biomedical Research: Progress and Potential
Introduction
On October 8, 2025, three scientists, Susumu Kitagawa, Richard Robson and Omar M. Yaghi, were awarded the Nobel Prize in Chemistry for their pioneering contributions to the field of metal-organic frameworks (MOFs). MOFs, as a major class of nanoporous materials, have attracted tremendous attention due to their tunable porosity, high surface areas, and versatile chemical functionalities, which offer unique opportunities for biomedical applications. Consequently, nanoporous materials remain at the forefront of research in materials science and biomedicine. Traditionally, nanoporous materials are a class of solid materials consisting of pores, or void spaces, with characteristic dimensions in the nanometer range (typically 1–100 nm). According to the International Union of Pure and Applied Chemistry (IUPAC) classification, nanopores can be categorized by pore diameter into micropores (<2 nm), mesopores (2–50 nm), and macropores (>50 nm).1 These pore size regimes critically determine application suitability. Microporous materials are generally suitable for small-molecule drug adsorption but exhibit limited capacity for macromolecular cargo. Mesoporous materials, such as mesoporous silica nanoparticles, offer optimal pore dimensions for protein, peptide, and nucleic acid loading, enabling high loading efficiency and tunable release kinetics. Macroporous structuresfacilitate enhanced mass transport and cell infiltration, making them more appropriate for tissue engineering and regenerative medicine applications. Owing to their high specific surface areas, tunable pore sizes and morphologies, and adjustable surface chemistries, nanoporous materials enable precise control over molecular transport and interfacial interactions. Consequently, they have been extensively explored for applications in catalysis,2 adsorption,3 separation,4 energy storage and conversion,5 as well as emerging biomedical fields such as drug delivery,6 biosensing,7 and tissue engineering.8 Despite these promising advances, regulatory approval pathways and clinical translation hurdles remain critical factors that strongly influence the successful implementation of nanoporous materials in biomedical practice. In addition to regulatory and clinical translation issues, challenges related to large-scale synthesis, reproducibility, and manufacturing scalability remain major obstacles for the practical application of nanoporous materials.
A large variety of nanoporous materials have been explored in biomedical fields including inorganic materials, organic materials, and hybrid materials according to their compositions.9 The unique physicochemical properties associated with compositions determine the performance and suitability for specific biomedical applications. For example, inorganic nanoporous materials such as mesoporous silica have been extensively applied in bone regeneration and tissue engineering owing to their excellent mechanical strength, chemical stability.10 In contrast, organic nanoporous materials, including covalent organic frameworks (COFs) and porous polymers, have shown great potential in drug delivery and biosensing, benefiting from their structural tunability, biocompatibility, and ease of functional modification.11 Moreover, hybrid nanoporous materials, such as MOFs, integrate the robustness of inorganic nodes with the versatility of organic linkers. Thus, MOFs have been explored for controlled drug release, photothermal therapy, and bioimaging, demonstrating synergistic performance that is difficult to achieve with single-component systems.12
To further enhance the physicochemical properties and expand their biomedical applications, morphology control and surface functionalization of nanoporous nanomaterials are crucial. Both the morphology and surface chemistry have significant impact on the interaction of nanoporous materials with either the active molecules as cargos, or biosystems (eg., serum proteins, cells with different types and phenotypes). These interactions not only determine the in vivo distribution and bio-interfacial behavior of the materials, but also directly affect their targeting efficiency. Consequently, the morphology control and surface functionalization provide useful strategy in passive and active targeting. Moreover, the pore size, composition, morphology and surface chemistry collectively affect other important properties of nanoporous materials, such as drug loading, controlled release, and biocompatibility. Table 1 provides a comparison of drug loading capacity and release kinetics across representative nanoporous materials. Due to variability in experimental conditions, biocompatibility is summarized qualitatively based on the literature (eg., “good” or “acceptable”).
 |
Table 1 Doxorubicin Loading Capacity and Release Kinetics Across Different Nanoporous Materials |
Rather than as absolute quantitative values. Recently, functionalized nanoporous materials have been applied far beyond controlled release, eg., in biosensing, imaging, and immunotherapy applications, highlighting their versatility as multifunctional platforms in modern biomedicine.
Despite numerous reviews focusing on structure classification, synthetic strategies, or specific biomedical applications of nanoporous materials,20–29 a systematic overview addressing the interplay between pore characteristics, surface functionalization, and biological interface interactions is still lacking. Furthermore, current literature rarely integrates discussions on multifunctional applications, translational challenges, and clinical relevance. To address these gaps, the present review categorizes nanoporous materials into inorganic, organic, and organic–inorganic hybrid classes, and further elucidates their interaction mechanisms at the biological interface, multifunctional biomedical applications, and translational considerations. This review covers key literature published between 1974 and 2025, with a primary focus on advances from 2018 onward to highlight recent developments in nanoporous materials and their biomedical applications. By providing a structured overview, this review aims to guide emerging researchers in the rational design of nanoporous materials for biomedical applications, bridging the gap between fundamental research and clinical translation.
Types of Nanoporous Materials Used in Biomedicine
Inorganic Nanoporous Materials
Inorganic nanoporous materials are among the most extensively studied due to their diverse structures, chemical stability, and tunable pore architectures. These include mesoporous silica, carbon-based materials, and metal oxides, etc. These materials are widely explored for biomedical applications.30
Mesoporous Silica
Mesoporous silica nanoparticles were first successfully synthesized and reported by the groups of Cai,31 Mann,32 and Ostafin.33 Subsequently, Victor Lin introduced the abbreviation “MSN” to denote mesoporous silica nanoparticles and facilitated their widespread use.34 Since then, MSNs with various morphologies, sizes, pore diameters, and pore structures have been synthesized and investigated. The morphological characteristics of MSNs influence their interactions with cells and tissues. Modifications such as PEGylation can reduce immune recognition and improve circulation time, while non-spherical morphologies may affect organ-specific accumulation. Zhao et al prepared virus-like MSNs, which, compared with conventional MSNs, exhibited unique internalization pathways, superior cellular uptake efficiency, and prolonged blood circulation time.35 Huang et al fabricated a series of MSNs with different morphologies and found that short-rod MSNs preferentially accumulated in the liver, whereas long-rod MSNs were more likely to enter the spleen. Moreover, PEGylation of these MSNs favored their distribution in the lungs (Figure 1).36 Li et al regulated the ratio of CTAB to rosin to prepare three types of MSNs with distinct morphologies, investigated their formation mechanisms, and identified MSN-80-PEG-FA as a promising nanoparticle for targeted drug delivery.37 Collectively, these studies demonstrate that MSN morphology can be rationally engineered to modulate cellular uptake pathways and organ-specific biodistribution; however, the in vivo behavior of non-spherical MSNs remains complex and is not yet fully predictable The size of nanoparticles is closely associated with cellular uptake and in vivo biodistribution. Mou et al reported that HeLa cells internalized MSNs with a diameter of 50 nm at a level approximately 2.5 times higher than that of 30 nm MSNs.38–40 Shi et al demonstrated that MSNs of various sizes predominantly accumulated in the liver and spleen, with smaller fractions distributed in the lungs, kidneys, and heart. Following intravenous injection for 30 minutes, the hepatic and splenic accumulation of MSNs increased with particle size from 80 to 120 and 200 nm. However, MSNs with a diameter of 360 nm exhibited a distinct distribution pattern in the spleen. Notably, smaller nanoparticles possessed a longer blood circulation time.41 Overall, these results indicate that particle size plays a decisive yet non-linear role in determining MSN biodistribution and circulation behavior, implying an inherent trade-off between cellular uptake efficiency and long term in vivo stability. The tunable pore size of MSNs is critical for accommodating molecules of different dimensions within the mesopores.42 To tailor pore sizes, swelling agents (eg., 1,3,5-trimethylbenzene,43 decane,44 hexane,45 and heptane)46 are often used to enlarge the micelles of the structure-directing agents, thereby yielding ordered mesoporous silica materials with larger pores. In addition, pore size can be further increased by adjusting the synthesis conditions,47–49 including extending the stirring time and elevating the hydrothermal treatment temperature,50 lowering the pH of the reaction system,51 or modifying the chain length of the surfactant.52,53 Such high tunability in pore size represents a key advantage of MSNs over many other nanoporous platforms, particularly for loading biomacromolecules and combination therapeutics. The pore structure type of MSNs also has a significant impact on their biomedical applications.Beyond classical MSNs such as MCM-41 and SBA-15, novel structures have been reported, including helical rod-like, core-shell dual-channel, radially mesoporous, and wormlike-pore MSNs.54–56 Zhang et al prepared hexagonal-pore MSNs, wormlike-pore MSNs, and enlarged-pore MSNs by varying the template agents and alkali sources, and synthesized HMSNs using a self-templating method. They subsequently modified the surfaces of hexagonal-pore and wormlike-pore MSNs with silane coupling agents or polydopamine, and found that hexagonal-pore MSN-NH2 and MSN-PDA served as anticancer drug delivery carriers with superior overall release performance.57 These examples highlight that pore architecture not only governs drug loading capacity but also critically influences release kinetics and therapeutic efficacy. Owing to their high surface area, tunable pore size and pore structure, and excellent biocompatibility, MSNs have been widely explored as drug delivery carriers, imaging agents, and platforms for multifunctional biomedical applications. Furthermore, their surface can be readily functionalized via versatile silanization chemistry, allowing for targeted delivery, controlled release, and integration with other therapeutic or diagnostic modalities. Nevertheless, limitations such as incomplete biodegradability and potential long-term accumulation should be carefully considered when evaluating MSNs in comparison with organic and composite nanoporous materials.
|
Figure 1 (a) Anticancer Doxorubicin (DOX) was used as a model drug to study effects of different types of MSN on drug loading and release. Reprinted with permission from Peng et al Copyright 2022 Elsevier.57 (b) Illustration of the fabrication of MSN-FITC. (c) TEM images of different shaped MSN-FITC. The shapes of particles were controlled by CTAB concentration. Reprinted with permission from Huang et al Copyright 2011 American Chemical Society.36 |
Metal and Metal Oxides
Since the successful synthesis of mesoporous silica was first reported in the 1990s, significant progress has been made in developing strategies to fabricate nanoporous materials using various components, such as metals and metal oxides.58,59 Compared with MSNs, nanoporous metals and metal oxides offer distinct advantages, including inherent structural stability, mechanical flexibility, and versatile surface functionalities, making them particularly attractive for industrial, biological, and analytical applications.29,60 Nanoporous gold (NPG) possesses a suitable pore size distribution and a large surface area, and due to the low coordination of gold atoms, it can significantly enhance the electrochemical responses toward enzyme substrates such as NADH and H2O2.61 Zhang et al developed a highly sensitive electrochemical DNA sensor by preparing an NPG electrode through the dealloying of Ag from an Au/Ag alloy and fabricating multifunctional encoded AuNPs (Figure 2). By leveraging the dual amplification effect of the NPG electrode and the multifunctional encoded AuNPs, this DNA biosensor enabled quantitative detection of DNA within the concentration range of 8.0 × 10−17 to 1.6 × 10−12 M (Figure 2).62 This example highlights how nanoporous metals can integrate structural and functional advantages for ultrasensitive detection, which is less readily achieved with conventional silica-based materials. Xu et al found that nanoporous oxidation of titanium alloys can generate nanopores and nanotubes on the surface, enhancing cell adhesion, proliferation, and matrix deposition, thereby promoting tissue growth and bone integration.63 Additionally, He et al fabricated titanium dioxide nanotubes via anodization, and the results showed that this nanotopography promoted macrophage recruitment, inhibited osteoclast activity, and regulated cytokine secretion through integrin signaling, ultimately improving the osteointegration of implants.64 These studies collectively indicate that metal and metal oxide nanoporous materials can provide both structural guidance and biofunctional cues, complementing the cargo-loading and delivery capabilities of MSNs. Recently, researchers have advanced the synthesis and functionalization of metal nanoparticles by precisely controlling their size, shape, and surface properties. These developments have greatly advanced metal nanoparticle synthesis and functionalization technologies. As a result, metal nanoparticles show enhanced potential in applications such as targeted drug delivery, gene therapy, biosensing, and tissue regeneration. In contrast, inorganic silica materials often require additional surface modification to achieve comparable functionality in these areas.
 |
Figure 2 Chronocoulometry Determination of DNA Hybridization through nanoporous gold electrode. Reprinted with permission from Hu et al Copyright 2008 American Chemical Society.62 (Red solid line: experimental chronocoulometric curve after DNA hybridization, Black dashed line: theoretical diffusion-controlled line (Anson equation)). |
Organic Nanoporous Materials
Organic nanoporous materials include covalent organic frameworks (COFs), conjugated microporous polymers (CMPs), and hyper-crosslinked polymers (HCPs). They offer structural tunability, biocompatibility, and high surface area for drug delivery, biosensing, and other biomedical applications.
Covalent Organic Frameworks (COFs)
COFs are crystalline porous polymers composed of light elements (C, H, O, N, B) linked by covalent bonds, including amide, azine, imine, boronate ester, hydrazone, and others.65,66. Since Yaghi’s first report in 2005,67 COFs have been extensively studies for their synthetic strategies, structural features, and diverse applications. Compared with inorganic nanoporous materials, COFs can reduce long-term toxicity risks due to their fully organic frameworks. Their intrinsic porosity, high surface area, and excellent biocompatibility make them promising candidates for diverse applications. In particular, their outstanding physicochemical properties and ultrathin architectures allow for enhanced cellular interactions and controlled release profiles, which are difficult to achieve with conventional inorganic platforms.57 Dong et al constructed a redox-responsive disulfide-linked porphyrin COF loaded with 5-fluorouracil (5-Fu). This COF can be effectively disassembled under the action of endogenous glutathione (GSH) in tumor cells, thereby achieving efficient release of 5-Fu for selective chemotherapy of tumor cells. Meanwhile, by combining GSH depletion-enhanced photodynamic therapy (PDT), the system realized synergistic ferroptosis-based therapy against MCF-7 breast cancer cells (Figure 3).68 Yan et al fabricated two three-dimensional COFs as carriers for drug delivery and investigated the relationship between drug loading/release rates and the pore size as well as the geometry of the COFs. The results demonstrated that 3D-PI-COFs exhibited a high drug loading capacity (>20%) and excellent drug release control (95%).69 In 2018, Bhaumik et al synthesized a two-dimensional COF (TrzCOF) with a nanorod-like structure, which could be directly employed as an anticancer drug for the treatment of colon cancer and exhibited high stability in biological media.70 Taken together, these examples demonstrate that COFs combine structural precision with functional versatility, allowing for stimuli-responsive drug release, high loading capacity, and enhanced therapeutic efficacy. Compared to MSNs and metal-based nanoporous materials, COFs offer the advantage of fully organic, covalently linked frameworks, which can reduce potential long-term toxicity while providing tunable porosity and functionality. However, challenges remain in large-scale synthesis and reproducibility, which are important considerations for clinical translation.
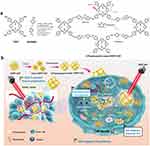 |
Figure 3 (a) Synthesis of DSPP-COF, nano DSPP-COF, and 5-Fu⊂nano DSPP-COF. (b) Synthesis and treatment application of 5-Fu⊂nano DSPP-COF, including GSH-promoted DSPP-COF nanocrystallization, 5-Fu loading, and the obtained 5-Fu⊂nano DSPP-COF for combination antitumor treatment by endogenous GSH-triggered drug release and GSH depletion-enhanced PDT via the ferroptosis pathway. Reproduced from Li et al Chemical science, 2023;14:1453–1460. Licensed under CC BY 3.0.68 The upward and downward arrows represent upregulation and downregulation, respectively. The “X” mark represents death. |
Conjugated Microporous Polymers (CMPs)
CMPs possess extended π-conjugated backbones and inherent porosity, thereby forming extended conjugated structures along their main chains.71 This unique architecture not only provides high surface area and tunable pore networks but also enables electronic and photophysical functionalities, which are difficult to achieve with conventional inorganic or purely organic nanoporous platforms. This combination of porosity and conjugation has promoted widespread applications of CMPs in the biomedical field, including biosensing, bioimaging, drug delivery, antibacterial therapy, and phototherapy.11,72–77 Kuo et al synthesized three different types of CMPs via Suzuki coupling condensation and investigated their physicochemical properties. The results revealed that TPE-Ph-BBT CMP exhibited the highest thermal stability. These CMPs can serve as effective carriers for tetracycline antibiotics, highlighting the great potential of CMPs in biomedical applications.78 Duan et al developed a novel cationic pyrimidine-modified conjugated microporous polymer (BPyMe-CMP), which exhibits excellent photoelectric effects. Under visible light irradiation, this CMP can generate reactive oxygen species (ROS) to eliminate bacteria, regulate macrophage polarization, promote angiogenesis, and accelerate cell migration, thereby facilitating wound healing (Figure 4).79 Taken together, CMPs combine intrinsic porosity with extended π-conjugation to provide a multifunctional platform capable of integrating drug delivery, antimicrobial activity, and tissue regeneration. Compared with MSNs and COFs, CMPs offer the additional advantage of inherent electronic/photophysical activity, enabling stimuli-responsive therapeutic or diagnostic functions without the need for external modification. However, limitations such as complex synthesis routes and potential scalability issues should be considered for practical biomedical applications.
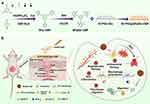 |
Figure 4 (a) Illustration of the synthesis of BPyMe-CMP and SF/PVA@BPyMe-CMP. (b) SF/PVA@BPyMe-CMP can accelerate the healing process of an infected wound by promoting fibroblast migration, killing bacteria, maintaining macrophage polarization and stimulating angiogenesis. Reprinted with permission from Liu et al Copyright 2024 Elsevier.79 The upward and downward arrows represent upregulation and downregulation, respectively. |
Hyper-Crosslinked Polymers (HCPs)
In the 1970s, Davankov’s research team pioneered the preparation of hyper-crosslinked polymers via post-crosslinking.80–82 HCPs are a prominent class of porous materials, characterized by large surface areas, remarkable chemical and thermal stability, tunable pore architectures, and facile preparation from low-cost monomers, making them attractive candidates for biomedical applications.83 Compared with other porous platforms such as MSNs, COFs, and CMPs, HCPs offer the advantages of low-cost synthesis, high stability, and permanent porosity, which can be easily functionalized for diverse biomedical applications. Their permanent porosity and chemical versatility enable the incorporation of diverse functional groups, thereby broadening their applicability in biosensing, drug delivery, and therapeutic platforms.84–86 For instance, Liu et al developed fluorescent hyper-cross-linked β-cyclodextrin-carbon quantum dot (β-CD-CQD) nanosponges with high biocompatibility and photoluminescence efficiency, serving as carriers for doxorubicin (DOX) to form DOX@β-CD-CQD nanomedicine. This system enables pH-responsive sustained release and targeted delivery within tumor cells, significantly enhancing antitumor efficacy and demonstrating promising potential for tumor theranostic applications.17 Moreover, Tan et al developed functional hypercrosslinked microporous polymer nanospheres (HMPNs) as effective drug delivery platforms for triple negative breast cancer (TNBC) treatment. The porous structure and high surface area of HMPNs enabled high loading of epirubicin (EPI) and pH-responsive controlled release, while their conjugated framework endowed strong blue fluorescence for bioimaging. Functionalization via imine chemistry and ssDNA modification further enhanced their versatility, demonstrating effective inhibition of TNBC cell proliferation and highlighting their potential in advanced biomedical applications (Figure 5).87 These studies illustrate that HCPs provide a highly versatile and robust platform for multifunctional biomedical applications, combining drug delivery, bioimaging, and stimuli-responsive capabilities. While HCPs share some features with CMPs and COFs, such as tunable porosity and functionalization potential, their simpler and lower-cost synthesis, together with exceptional chemical and thermal stability, offers a distinct advantage for scalable biomedical applications. Nevertheless, challenges remain in achieving precise structural control at the nanoscale compared to crystalline frameworks like COFs.
 |
Figure 5 (a) Synthetic scheme of HMPNs, HMPNs-EDA and HMPNs-EDA-ssDNA. (b) SEM micrographs of i) HMPNs ii) HMPNs-EDA iii) HMPNs-EDA-ssDNA (scale bar, 1 µm) and TEM images iv) HMPNs v) HMPNs-EDA vi) HMPNs-EDA-ssDNA, (scale bar, 200 nm). Reprinted with permission from Razzaque S. et al Copyright 2022 Elsevier.87 |
Hybrid Nanoporous Materials
Hybrid nanoporous materials represent a unique class of porous systems that integrate both inorganic and organic components, thereby combining the mechanical stability and tunable pore structures of inorganic frameworks with the chemical versatility and functionalizability of organic moieties. Representative examples include metal-organic frameworks (MOFs), Covalent inorganic-organic hybrid frameworks (CIOFs), and functionalized mesoporous silica materials.
Metal-Organic Frameworks (MOFs)
Metal-organic frameworks (MOFs) are an emerging class of hybrid materials that bridge inorganic chemistry and polymer science. By integrating inorganic metal nodes with organic linkers, MOFs combine the advantages of both inorganic stability and organic tunability, enabling highly versatile structural and functional properties that distinguish them from purely inorganic or purely organic nanoporous materials. Their tunable structural design endows these materials with versatile functionalities, including gas storage, catalysis, sensing, and biomedical applications.88 MOFs achieve exceptional structural tunability through the rational selection of metal nodes and organic linkers, enabling customized architectures and functionalities that accelerate their expanding role in biomedical applications.89–92 In particular, their modularity allows precise control over pore size, surface chemistry, and degradation behavior, making MOFs uniquely suited for targeted drug delivery and stimulus-responsive therapeutic platforms. He et al developed a chitosan composite scaffold (CS/DOX@Ti-MOF) that combines tumor therapy with bone regeneration. The scaffold enables efficient DOX loading and stimulus-responsive release to eliminate residual tumor cells, while simultaneously promoting osteogenic differentiation and bone defect repair, offering a promising strategy for postoperative bone tumor treatment.93 Moreover, Zheng et al developed a therapeutic nanoplatform based on Mg-MOF as the core. This platform undergoes gradual degradation during bone healing, releasing bioactive products that modulate the repair microenvironment, such as magnesium ions that facilitate angiogenesis.94 However, the potential long-term accumulation of magnesium ions in vivo could affect systemic mineral homeostasis and other biological processes, and therefore warrants careful evaluation of systemic exposure, biocompatibility, and overall safety. Overall, these examples illustrate that MOFs provide a highly versatile and customizable platform for biomedical applications, combining targeted drug delivery, controlled release, and regenerative capabilities. While MOFs provide a unique combination of structural precision, tunable biodegradability, and hybrid functionality, their long-term biocompatibility and scalability for large-scale production still require careful evaluation.
Covalent Inorganic-Organic Hybrid Frameworks (CIOFs)
Compared with covalent organic frameworks, CIOFs formed by covalent linkage of inorganic heterocycles and organic units, remain a relatively untapped and highly promising field of study.95 By integrating inorganic and organic components at the molecular level, CIOFs can combine the chemical robustness of inorganic motifs with the structural tunability and functional versatility of organic units, offering unique physicochemical properties that are difficult to achieve with purely organic or inorganic frameworks. Sun et al sythesized FeOx@SPNO-C core-shell nanospheres using cyclotriphosphazene-derived COIFs and Fe3O4. The catalyst showed excellent PMS-activated degradation of sulfamethoxazole, with high removal efficiency, low iron leaching, and enhanced reaction kinetics. The improved performance was attributed to synergistic FexO-SPNO-C interactions, abundant structural defects, sp2 carbon with C=O groups, and N/Fe-Nx active sites, highlighting its potential as a commercial Fenton-like catalyst.96 Tan et al conducted a long-term field trial to evaluate the effects of organic-inorganic compound fertilizers (COIFs) on summer maize growth, yield, nutrient utilization, and soil quality. COIF applied at 90% of the recommended rate (COIF1) enhanced biomass, grain yield, nutrient use efficiency, and soil quality compared to conventional chemical fertilizers, while improving soil organic matter, nutrient availability, and pH. The study demonstrates that COIFs are an effective strategy to reduce chemical fertilizer use, improve soil health, and boost maize productivity.97 Although biomedical applications of CIOFs are currently limited, their combination of inorganic and organic characteristics suggests considerable potential for drug delivery, imaging, and other therapeutic platforms. In particular, the inherent tunability, functional group diversity, and structural robustness of CIOFs may enable the design of hybrid nanoplatforms that overcome some limitations of purely organic (COFs, CMPs) or inorganic (MSNs, HCPs) nanoporous materials.
Organic Functionalized Mesoporous Silica Nanoparticles (OFMSNs)
OFMSNs obtained by incorporating organic moieties into mesoporous silica frameworks, have emerged as versatile platforms for diverse applications. The introduction of organic functionalities allows fine-tuning of surface chemistry, enhancing stability, selective adsorption, and stimuli-responsive behavior, which complement the inherent structural features of conventional MSNs.98 For example, Gianotti et al prepared amino-functionalized mesoporous silica nanoparticles via post-synthesis grafting and co-condensation strategies. The spatial distribution of amino groups was assessed through the aggregation behavior of a grafted photosensitizer (Verteporfin) using spectroscopic analysis. Furthermore, the authors investigated the formation of a protein corona on both bare and amino-modified mesoporous silica by employing bovine serum albumin as a model protein.99 In another study, Lin et al developed phenanthridinium-functionalized mesoporous silica nanoparticles with tunable surface localization, demonstrating that exterior decoration induced strong cytotoxicity via mRNA binding in HeLa cells, while interior functionalization significantly reduced toxicity (Figure 6).100 These findings illustrate that surface organic functionalization extends the functionality of MSNs, enabling precise control over biological interactions, cytotoxicity, and protein adsorption. Compared with unmodified MSNs, OFMSNs offer enhanced versatility for biomedical applications, while maintaining the structural robustness and tunable porosity of the silica framework. However, careful design is required to balance functionalization density and biological safety, as excessive or improperly localized functional groups may induce cytotoxicity or undesired protein interactions.
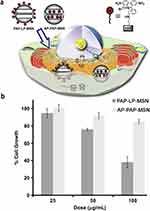 |
Figure 6 (a) Schematic representation of the endocytosis of AP-PAP-MSN/PAP-LP-MSN into HeLa cells and binding to cytoplasmic oligonucleotides. The blue arrows indicate the intracellular transport pathway of the nanoparticles leading to their interaction with cytoplasmic oligonucleotides. (b) Cell growth inhibition of HeLa as a function of the concentration of AP-PAP-MSN (light-grey bars), and PAP-LP-MSN (dark-grey bars). Reprinted with permission from Vivero-Escoto JL et al Copyright 2010 Elsevier.100 |
Mechanisms of Interaction Between Nanoporous Materials and Biological Systems
The interaction mechanisms between nanomaterials and biological systems are of great significance for the rational design and development of nanomaterials. As a class of nanomaterials with considerable potential for biomedical applications, elucidating the interaction mechanisms of nanoporous materials with biological systems is of profound importance.
Mechanisms at the Molecular and Ionic Level
At the molecular and ionic level, the interaction between nanoporous materials and biological systems is primarily governed by non-covalent forces,101 electrostatic interactions,102 and coordination chemistry.103 Importantly, these interactions are strongly regulated by nanoporosity-related parameters, including pore size, pore geometry, surface charge distribution, and nanoscale confinement effects. The high surface area and tunable surface chemistry of nanoporous materials enable strong adsorption of biomolecules, such as proteins, nucleic acids, and small-molecule drugs. Electrostatic attraction or repulsion between charged pore surfaces and ionic species in the biological environment can significantly influence protein folding, conformational stability, and enzymatic activity. Moreover, the confinement effect within nanoscale pores can alter the hydration shell of biomolecules, modulate hydrogen bonding, and even stabilize otherwise unstable conformers. For drug molecules, ionic interactions and pore confinement contribute to enhanced stability, controlled release, and protection against enzymatic degradation. These nanoporosity-driven molecular and ionic mechanisms constitute the fundamental physicochemical basis linking pore structure to downstream biological responses. For example, the integration of silica, titania, and carbon-based nanomaterials, along with PEGylation, further improves their bioactivity, reduces immune responses, and prolongs circulation time.104,105 Moreover, the adjustable mechanical properties of nanoporous scaffolds make them highly promising for tissue engineering and regeneration.106
Mechanisms at the Cell Level
At the cellular level, the interaction between nanoporous materials and biological systems is largely determined by cellular uptake pathways, membrane interactions, and intracellular trafficking. Notably, protein corona formation, a key determinant of nanomaterial biological identity, is strongly regulated by the pore architecture, surface porosity, surface charge, and chemical functionality of nanoporous materials. These physicochemical properties modulate the composition, conformation, and stability of the adsorbed protein corona, thereby governing particle-cell recognition and internalization efficiency. Pore size and connectivity influence protein accessibility and spatial confinement, where mesoporous structures can enhance protein adsorption and retention, while nanoscale surface roughness and curvature may induce protein conformational rearrangements that alter downstream cellular responses. Caruso et al investigated the distinct effects of protein coronas formed by proteins adsorbed onto nanoporous polymers on particle-cell interactions, highlighting the correlation between the in situ conformation of the protein corona, the associated biological properties, and the resulting biological functions.107 Nanoporous materials can be internalized via endocytosis, phagocytosis, or micropinocytosis depending on their size, shape, surface chemistry, and porosity. Porous surfaces can promote membrane ruffling and macropinocytosis by increasing local membrane curvature and adsorption density of membrane-associated proteins. Persson et al demonstrated that nanomaterials promote cellular uptake by enhancing the delivery of lentiviruses, baculoviruses, mRNA, antibodies, and liposomes, and further identified macropinocytosis as the primary mechanism of nanoporous surface-induced uptake through inhibition of individual endocytic pathways (Figure 7).108 In addition, particle size, shape, and surface charge critically modulate uptake mechanisms: smaller nanoparticles typically favor clathrin-mediated endocytosis, whereas larger or elongated particles are more prone to macropinocytosis or phagocytosis. Surface functionalization, such as ligand conjugation, can trigger receptor mediated endocytosis, while the formation of a protein corona can further bias cellular recognition and internalization routes. Mechanical properties and nanoscale surface topography also contribute by affecting membrane deformation and cytoskeletal rearrangements required for specific uptake pathways. Once internalized, pore size, connectivity, and surface functionality regulate intracellular release kinetics and bioavailability of encapsulated biomolecules. Surface modifications, such as PEGylation or ligand conjugation, can enhance cellular targeting, reduce non-specific uptake, and indirectly reshape protein corona composition, thereby modulating immune recognition.109 Additionally, the mechanical properties and nanoscale topography of porous scaffolds can affect cell adhesion, spreading, and differentiation, supporting tissue regeneration. Bernotienė et al highlighted in the progress of stem cells and scaffolds in regenerative medicine that the mechanical properties, biodegradability, and pore size of scaffolds play crucial roles in nutrient exchange, cell adhesion, migration, and differentiation, and are key factors for achieving effective tissue repair.110 Together, these cellular-level mechanisms complement the molecular and ionic interactions, providing a comprehensive understanding of how nanoporous materials elicit controlled biological responses.
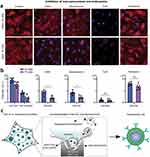 |
Figure 7 Mechanism of enhanced uptake on porous surfaces. (a) Confocal images of Rhodamine B-Dextran (10 or 70 kDa) uptake by mouse pre-osteoblasts on porous (400 nm) PCL films (scale bar: 5 µm); nuclei stained with Hoechst. Cells were pre-treated with inhibitors (EIPA, Wortmannin, CyD, Dynasore). (b) Quantification of Rhodamine B-Dextran uptake by plate reader, showing inhibition effects. (c) Schematic of nanotopography-induced micropinosome formation and downstream cellular processes. Reproduced from Aramesh M. et al Advanced Functional Materials, 34(28), 2,400,487 (2024), licensed under CC BY-NC-ND 4.0.108 A two-way ANOVA test was conducted to assess the statistical significance (*:p < 0.05). “ns” stands for “ not significant”. |
Mechanisms at the Tissue and System Level
At the tissue and system level, the effects of nanoporous materials are governed by their interactions with extracellular matrices, tissue architecture, and systemic physiological processes.111 Hierarchical porosity and interconnected pore networks enable nanoporous scaffolds to mimic native tissue microenvironments, facilitating cell infiltration, vascularization, and extracellular matrix remodeling. Lee et al proposed a low-temperature sintering method to fabricate hydroxyapatite (HAP) scaffolds with 30 nm pores that mimic bone morphology. These nanopores enhanced the proliferation and differentiation of preosteoblasts and promoted bone regeneration by improving water and protein adsorption, highlighting their potential as a novel substitute for bone repair.112 Such examples highlight how nanoscale pore dimensions directly translate into improved tissue-level biological outcomes. The controlled release of bioactive molecules from these materials can modulate local tissue responses, including angiogenesis, immune modulation, and extracellular matrix remodeling (Figure 8).113 Systemically, pore size, particle dimensions, and surface chemistry collectively determine biodistribution, circulation time, and clearance behavior, thereby influencing therapeutic efficacy and safety.114 Integration of multifunctional modifications, such as PEGylation and targeting ligands, can further optimize tissue-specific delivery, reduce off-target effects, and enhance overall biocompatibility.115 These tissue- and system-level mechanisms, together with molecular, ionic, and cellular interactions, provide a holistic framework for understanding and designing nanoporous materials for advanced biomedical applications.
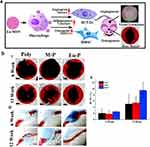 |
Figure 8 Eu-MSNs enhance cranial bone regeneration and neovascularization. (a) Eu-MSNs stimulated macrophages, which promoted osteogenic differentiation of BMSCs (upregulation of COL-I, OCN, ALP, RUNX2) and angiogenic differentiation of HUVECs (upregulation of CD31, MMP9, VEGFR, PDGFR). Subsequent in vivo experiments showed enhanced cranial bone repair and neovascularization at chronic wound sites. (b) Effect of Eu-MSNs on cranial defect osteogenesis: (i) Representative Micro-CT images of new bone formation at 6 and 12 weeks (grey: normal skull; black: defect; red: newly formed bone; analyzed with CTAn). (ii) Corresponding quantitative analysis showing larger new bone area in Eu-MSNs-Polymer (Eu-P) group compared with Polymer (Poly) and MSNs-Polymer (M-P) groups. (iii) VG-stained immunofluorescence images of cranial defect cross-sections at 6 and 12 weeks confirm Micro-CT results, showing increased bone formation in Eu-P groups. Reprinted with permission from Shi et al Copyright 2017 Elsevier.113 (i) scale bar = 1 mm, (iii) scale bar = 1 mm. The red arrows represent upregulation. |
Biomedical Application
Intelligent Nanoporous Materials for Cancer Therapy
The pathological hallmarks of diseases have motivated the rational design of stimuli-reponsive nanoporous carriers. For example, based on the acidic conditions, overexpressed glutathione, and hypoxia commonly found in tumors, researchers have designed intelligent nanoporous drug delivery systems with responsive behaviors. Zhou et al developed a pH-responsive gatekeeper by coating mesoporous silica nanoparticles with Eu-GMP metal-organic frameworks, enabling controlled release of both antigens and adjuvants. The MSN-OVA@MOF@CpG system effectively delivered ovalbumin and CpG to antigen-presenting cells, leading to enhanced T cell responses and efficient suppression of melanoma growth and metastasis.116 Gong et al developed a GSH-responsive silica nanoparticle capable of delivering mRNA and ribonucleoprotein precisely to retinal pigment epithelium and liver cells, achieving efficient genome editing and demonstrating great potential for applications in gene therapy and genome editing (Figure 9).117 While these studies focus on pH- and GSH-responsive systems, emerging stimuli such as magnetic field-responsive,118,119 and ultrasound-responsive platforms120 provide additional strategies for precise spatiotemporal control of therapeutic delivery. Yang et al designed and fabricated a dual-responsive soft robot based on pNIPAM and MoS2, which utilizes magnetic and photothermal responsiveness to achieve high flexibility, controllable motion, and targeted drug release, demonstrating potential clinical applicability in a gastric model.121 These studies highlight the promising potential of environment-responsive nanoporous materials for precise, controlled, and effective therapeutic delivery in various biomedical applications.
 |
Figure 9 (a) Illustration of the multifunctional SNP for the delivery of nucleic acids (eg., DNA and mRNA) and CRISPR genome editor (eg., RNP, RNP + ssODN). (b) Efficient delivery of mRNA by SNP-PEG-ATRA via local administration in mouse retinal pigment epithelium (RPE), and by SNP-PEG-GalNAc via intravenous injection in liver tissue (upper panel); Efficient delivery of CRISPR-Cas9 RNP by SNP-PEG-ATRA via local administration in mouse RPE, and by SNP-PEG-GalNAc via intravenous injection in liver tissue (lower panel). Reprinted with permission from Wang et al Copyright 2021 Elsevier.117 |
Tissue Repair and Regeneration
Tissue repair occurs after injury through processes such as inflammation, fibroblast proliferation, collagen deposition, and scar formation, thereby restoring tissue integrity and basic functions. Tissue regeneration, in contrast, refers to the replacement of damaged tissues or organs with newly formed ones that are structurally and functionally similar to the original. Tissue repair and regeneration help maintain homeostasis and reduce functional loss, and they form the foundation for the development of regenerative medicine and tissue engineering. Nanoporous materials, owing to their unique properties such as high surface area, tunable porosity, and excellent biocompatibility, hold great potential for applications in tissue repair and regeneration. For example, silica nanoparticles can enhance bone regeneration by increasing the solubility of therapeutic agents, thereby improving their bioavailability and ultimately augmenting the therapeutic efficacy of the encapsulated drugs in bone regeneration applications.122,123 MOFs, due to their highly ordered, porous, and three-dimensional crystalline structures, can serve as biomaterials in tissue engineering for orthopedic implants, cardiovascular devices, neural tissue engineering, and skin wound care.124 Antithrombotic drugs and small-molecule nitric oxide donors are often used to dilate blood vessels and prevent arterial occlusion.125 Reynolds et al demonstrated that the copper-based MOF Cu-BTC is an effective catalyst for converting S-nitrosocysteine in the blood into nitric oxide and cysteine, and in vitro studies showed that it exhibits good hemocompatibility.126 Together, these examples underscore the significant potential of nanoporous materials, including silica nanoparticles and MOFs, as versatile platforms for enhancing tissue repair and regeneration. However, degradation rates and degradation byproducts can vary substantially across inorganic, organic, and hybrid nanoporous materials, and these factors should be carefully considered when evaluating long-term biocompatibility and in vivo safety.
Theranostics
Theranostics, which integrates diagnostic and therapeutic functions into a single nanosystem, holds great potential in advancing personalized medicine in the biomedical field.127 Beyond drug delivery, nanoporous materials can also serve as theranostic platforms by co-loading therapeutic agents and imaging or sensing probes, enabling real-time monitoring of drug distribution and therapeutic efficacy. Nanoporous materials impart unique properties that enable the integration of various types of drugs, biomarkers, fluorophores, and linkers into a single delivery system, thereby realizing theranostics. Cai et al reported a novel ultrasmall porous silica nanoparticle conjugated with the isotopic pair86,90 Y, enabling the combination of noninvasive imaging and internal radiotherapy. The results demonstrated that86 Y-DOTA-UPSN prolonged blood circulation and provided excellent tumor imaging contrast, while90 Y-DOTA-UPSN significantly inhibited tumor growth and extended overall survival, highlighting its potential for clinical translation as a cancer theranostic candidate (Figure 10).128 Zhang et al used PCN-600 to load antitumor drugs and polydopamine (PDA). The ligand tetrakis(4-carboxyphenyl)porphyrin (TCPP) in PCN-600 can generate singlet oxygen (1O2) to kill tumor cells, while PDA serves as a photothermal agent for photothermal therapy (PTT). The central Fe3⁺ ion in PCN enables tumor diagnosis and treatment through magnetic resonance imaging (MRI)-guided chemotherapy combined with photothermal and photodynamic synergistic therapy.129
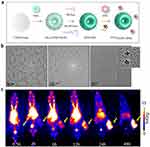 |
Figure 10 (a) Schematic representation of the UPSN synthesis method. (b) Transmission electron microscopy (TEM) images of UPSN at different magnifications. The inset green box corresponds to a magnified image of individual nanoparticles. (c) Serial MIP PET images of 4T1 tumor-bearing mice injected with 86Y-DOTA-UPSN (n = 5). Tumors are indicated with yellow arrows. Reprinted with permission from Chen et al Copyright 2023 American Chemical Society.128 |
Conclusion and Outlook
In summary, the structural tunability, large surface area, controllable porosity, and versatile chemical functionalities of nanoporous materials make them a highly promising class of nanomaterials for diverse biomedical applications. As summarized in this review, diverse types of nanoporous materials-including inorganic, organic, and hybrid systems-exhibit distinct advantages in biomedicine, ranging from intelligent drug and gene delivery to tissue repair, immunomodulation, and integrated theranostic platforms. In particular, inorganic nanoporous materials (eg., mesoporous silica and metal oxides) provide robust structural support and controllable pore architectures, organic nanoporous materials (eg., COFs, CMPs, and HCPs) offer high chemical tunability and potential biodegradability, while hybrid systems such as MOFs and functionalized silica combine structural precision with multifunctionality. By integrating structure–property–biointerface relationships, this review moves beyond descriptive summaries to provide a mechanism-oriented framework for rational material design. Mechanistic investigations at the molecular, cellular, and tissue levels have further elucidated how nanoporous materials interact with biological systems, providing critical insights for the rational design of safe, effective, and multifunctional nanoplatforms.
Despite these remarkable advances, several challenges remain before the widespread clinical translation of nanoporous materials can be realized. Key unresolved issues include potential toxicity, long-term stability and degradation behavior in vivo, immune recognition and clearance, as well as batch-to-batch reproducibility and large-scale manufacturability. First, precise control over synthesis, reproducibility, and scalability is required to ensure consistent physicochemical properties and biological performance. Second, a deeper understanding of long-term biocompatibility, biodegradation, and potential immunogenicity is essential for ensuring safety in vivo. Third, immune evasion and controlled interactions with the protein corona remain critical design considerations that directly influence circulation time and therapeutic efficacy. These limitations highlight critical gaps that must be addressed before meaningful clinical translation can occur. Finally, regulatory pathways and realistic timeline expectations for clinical translation, including preclinical validation and phased clinical trials, should be acknowledged to guide research priorities. Interdisciplinary approaches that combine materials science, biology, and clinical medicine will be critical for translating nanoporous materials from laboratory research to clinical biomedical applications.
Looking forward, emerging strategies such as AI-driven computational design, bio-inspired nanoporous architectures, and scalable manufacturing methods are expected to further accelerate the development of advanced nanoporous biomaterials. AI and machine learning can facilitate rational design of pore structures, surface chemistry, and stimuli-responsive behaviors for optimized biomedical performance. Bio-inspired scaffolds can mimic natural tissue architectures, improving cell adhesion, proliferation, differentiation, and dynamic responsiveness. Scalable and reproducible manufacturing methods, including template-assisted assembly, continuous-flow synthesis, and 3D printing, will enable reliable production of multifunctional nanoplatforms suitable for preclinical and clinical studies.
Collectively, integrating these emerging trends with current knowledge of molecular, cellular, and tissue-level interactions will pave the way for the next generation of intelligent, adaptive, and personalized nanoporous biomaterials. Accordingly, future research should prioritize hypothesis-driven validation in disease-specific models, standardized protocols for scalable synthesis, and systematic biosafety evaluation to ensure translational readiness. With continued advances in nanotechnology, bioengineering, and computational modeling, nanoporous materials are poised to play a transformative role in precision medicine, regenerative therapy, and next-generation theranostics, ultimately bridging the gap between nanomaterial innovation and clinical application.
Acknowledgments
We acknowledge the support from The University of Queensland, the China Scholarship Council (202308410528), the Australian Research Council and National Health and Medical Research Council.
Disclosure
The authors report no conflicts of interest in this work.
References
1. Thommes M, Kaneko K, Neimark AV, et al. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem. 2015;87(9–10):1051–22. doi:10.1515/pac-2014-1117
2. Li Q, Kudo A, Ma J, et al. Tuning electrocatalytic activities of dealloyed nanoporous catalysts by macroscopic strain engineering. Nano Lett. 2024;24(18):5543–5549. doi:10.1021/acs.nanolett.4c01469
3. Manitsas L, Fanourgakis GS. A physically motivated Machine Learning model for accurate gas adsorption predictions in nanoporous materials. Microporous Mesoporous Mater. 2025;398:113796. doi:10.1016/j.micromeso.2025.113796
4. Schlaich A, Barrat J-L, Coasne B. Theory and modeling of transport for simple fluids in nanoporous materials: from microscopic to coarse-grained descriptions. Chem Rev. 2025;125(5):2561–2624. doi:10.1021/acs.chemrev.4c00984
5. Davidraj JM, Sathish CI, Benzigar MR, et al. Recent advances in food waste-derived nanoporous carbon for energy storage. Sci Technol Adv Mater. 2024;25(1):2357062. doi:10.1080/14686996.2024.2357062
6. Zhang R, Hua M, Liu H, Li J. How to design nanoporous silica nanoparticles in regulating drug delivery: surface modification and porous control. Mater Sci Eng B. 2021;263:114835. doi:10.1016/j.mseb.2020.114835
7. Xiao S, Wang S, Wang X, Xu P. Nanoporous gold: a review and potentials in biotechnological and biomedical applications. Nano Select. 2021;2(8):1437–1458. doi:10.1002/nano.202100006
8. Maksoud FJ, De la paz MFV, Hann AJ, et al. Porous biomaterials for tissue engineering: a review. J Mater Chem B. 2022;10(40):8111–8165. doi:10.1039/D2TB01242E
9. Zhao N, Yan L, Zhao X, et al. Versatile types of organic/inorganic nanohybrids: from strategic design to biomedical applications. Chem Rev. 2018;119(3):1666–1762. doi:10.1021/acs.chemrev.8b00201
10. Sanchez C, Boissiere C, Cassaignon S, et al. Molecular engineering of functional inorganic and hybrid materials. Chem Mater. 2014;26(1):221–238. doi:10.1021/cm402528b
11. Zhu Y, Xu P, Zhang X, Wu D. Emerging porous organic polymers for biomedical applications. Chem Soc Rev. 2022;51(4):1377–1414. doi:10.1039/D1CS00871D
12. Sanchez C, Belleville P, Popall M, Nicole L. Applications of advanced hybrid organic-inorganic nanomaterials: from laboratory to market. Chem Soc Rev. 2011;40(2):696–753. doi:10.1039/C0CS00136H
13. Bhattarai JK, Neupane D, Nepal B, Demchenko AV, Stine KJ. Nanoporous gold monolith for high loading of unmodified doxorubicin and sustained co-release of doxorubicin-rapamycin. Nanomaterials. 2021;11(1):208. doi:10.3390/nano11010208
14. Ortiz AC, Jackson N, Buelvas N, et al. Release kinetics approach of stimuli-responsive mesoporous silica nanocarriers: pH-sensitive linker versus pH-sensitive framework. J Drug Delivery Sci Technol. 2024;91. 105212. doi:10.1016/j.jddst.2023.105212
15. Chen P, Li Y, Dai Y, et al. Porphyrin-based covalent organic frameworks as doxorubicin delivery system for chemo-photodynamic synergistic therapy of tumors. Photodiagn Photodyn Ther. 2024;46:104063. doi:10.1016/j.pdpdt.2024.104063
16. Zhang M, Zhang S, Zhang K, et al. Self-assembly of polymer-doxorubicin conjugates to form polyprodrug micelles for pH/enzyme dual-responsive drug delivery. Colloids Surf A. 2021;622:126669. doi:10.1016/j.colsurfa.2021.126669
17. Pei M, Pai J-Y, Du P, Liu P. Facile synthesis of fluorescent hyper-cross-linked β-cyclodextrin-carbon quantum dot hybrid nanosponges for tumor theranostic application with enhanced antitumor efficacy. Mol Pharm. 2018;15(9):4084–4091. doi:10.1021/acs.molpharmaceut.8b00508
18. Aljazzar SO. Encapsulation of the anticancer drug doxorubicin within metal-organic frameworks: spectroscopic analysis, exploration of anticancer, antioxidant, antibacterial properties, and optimization through Box-Behnken design. J Indian Chem Soc. 2024;101(3):101125. doi:10.1016/j.jics.2024.101125
19. Day CM, Sweetman MJ, Song Y, Plush SE, Garg S. Functionalized mesoporous silica nanoparticles as delivery systems for doxorubicin: drug loading and release. Appl Sci. 2021;11(13):6121. doi:10.3390/app11136121
20. Parvin N, Kumar V, Mandal TK, Joo SW. Advancements in nanoporous materials for biomedical imaging and diagnostics. J Funct Biomater. 2024;15(8):226. doi:10.3390/jfb15080226
21. Adiga SP, Curtiss LA, Elam JW, et al. Nanoporous materials for biomedical devices. JOM. 2008;60(3):26–32. doi:10.1007/s11837-008-0014-2
22. Orellano MS, Sanz O, Camarero-Espinosa S, Beloqui A, Calderon M. Recent advances and future perspectives of porous materials for biomedical applications. Nanomedicine. 2022;17(4):197–200. doi:10.2217/nnm-2021-0300
23. Yapa P, Munaweera I. Functionalized nanoporous architectures derived from sol–gel processes for advanced biomedical applications. J Mater Chem B. 2025;13(5):1092–1110. doi:10.1039/D4TB02568A
24. Lei Q, Guo J, Noureddine A, et al. Sol–gel-based advanced porous silica materials for biomedical applications. Adv Funct Mater 2020;30(41):1909539. doi:10.1002/adfm.201909539
25. Radhakrishnan D, Mohanan S, Choi G, et al. The emergence of nanoporous materials in lung cancer therapy. Sci Technol Adv Mater. 2022;23(1):225–274. doi:10.1080/14686996.2022.2052181
26. Wu KY, Brister D, Bélanger P, Tran SD. Exploring the potential of nanoporous materials for advancing ophthalmic treatments. Int J Mol Sci. 2023;24(21):15599. doi:10.3390/ijms242115599
27. Głowniak S, Szczęśniak B, Choma J, Jaroniec M. Advances in microwave synthesis of nanoporous materials. Adv Mater. 2021;33(48):2103477. doi:10.1002/adma.202103477
28. Zhang L, Jaroniec M. Strategies for development of nanoporous materials with 2D building units. Chem Soc Rev. 2020;49(13):4203–4221. doi:10.1039/D0CS00185F
29. Hadden M, Martinez-Martin D, Yong K-T, Ramaswamy Y, Singh G. Recent advancements in the fabrication of functional nanoporous materials and their biomedical applications. Materials. 2022;15(6):2111. doi:10.3390/ma15062111
30. Zheng H, Gao F, Valtchev V. Nanosized inorganic porous materials: fabrication, modification and application. J Mater Chem A. 2016;4(43):16756–16770. doi:10.1039/C6TA06441D
31. Cai Q, Luo Z-S, Pang W-Q, Fan Y-W, Chen X-H, Cui F-Z. Dilute solution routes to various controllable morphologies of MCM-41 silica with a basic medium. Chem Mater. 2001;13(2):258–263. doi:10.1021/cm990661z
32. Fowler C, Khushalani D, Lebeau B, Mann S. Nanoscale materials with mesostructured interiors. Adv Mater. 2001;13(9):649–652. doi:10.1002/1521-4095(200105)13:9
33. Nooney RI, Thirunavukkarasu D, Chen Y, Josephs R, Ostafin AE. Synthesis of nanoscale mesoporous silica spheres with controlled particle size. Chem Mater. 2002;14(11):4721–4728. doi:10.1021/cm0204370
34. Lai C-Y, Trewyn BG, Jeftinija DM, et al. A mesoporous silica nanosphere-based carrier system with chemically removable CdS nanoparticle caps for stimuli-responsive controlled release of neurotransmitters and drug molecules. J Am Chem Soc. 2003;125(15):4451–4459. doi:10.1021/ja028650l
35. Dong Y, Lu B, Zang S, Zhao J, Wang X, Cai Q. Removal of methylene blue from coloured effluents by adsorption onto SBA-15. J Chem Technol Biotechnol. 2011;86(4):616–619. doi:10.1002/jctb.2558
36. Huang X, Li L, Liu T, et al. The shape effect of mesoporous silica nanoparticles on biodistribution, clearance, and biocompatibility in vivo. ACS Nano. 2011;5(7):5390–5399. doi:10.1021/nn200365a
37. Yang W, Song FX, Wang S, Zhang L, Zeng X, Li Y. Multifunctional mesoporous silica nanoparticles with different morphological characteristics for in vitro cancer treatment. Colloids Surf A. 2021;610:125717. doi:10.1016/j.colsurfa.2020.125717
38. Lu F, Wu S-H, Hung Y, Mou C-Y. Size effect on cell uptake in well-suspended, uniform mesoporous silica nanoparticles. Small. 2009;5(12):1408–1413. doi:10.1002/smll.200900005
39. Chithrani BD, Ghazani AA, Chan WC. Determining the size and shape dependence of gold nanoparticle uptake into mammalian cells. Nano Lett. 2006;6(4):662–668. doi:10.1021/nl052396o
40. Osaki F, Kanamori T, Sando S, Sera T, Aoyama Y. A quantum dot conjugated sugar ball and its cellular uptake. On the size effects of endocytosis in the subviral region. J Am Chem Soc. 2004;126(21):6520–6521. doi:10.1021/ja049043i
41. He Q, Zhang Z, Gao F, Li Y, Shi J. In vivo biodistribution and urinary excretion of mesoporous silica nanoparticles: effects of particle size and PEGylation. Small. 2011;7(2):271–280. doi:10.1002/smll.201001459
42. Douroumis D, Onyesom I, Maniruzzaman M, Mitchell J. Mesoporous silica nanoparticles in nanotechnology. Crit Rev Biotechnol. 2013;33(3):229–245. doi:10.3109/07388551.2012.685960
43. Suteewong T, Sai H, Cohen R, et al. Highly aminated mesoporous silica nanoparticles with cubic pore structure. J Am Chem Soc. 2011;133(2):172–175. doi:10.1021/ja1061667
44. Zhang H, Shen Y. Straight chain alkane-assisted synthesis of mesoporous silica. Mater Lett. 2012;75:183–185. doi:10.1016/j.matlet.2012.02.026
45. Shan W, Wang W, Ru H. Siliceous mesocellular foams modified via a partitioned cooperative self-assembly process using hexane as pore swelling agent. J Non-Cryst Solids. 2015;425:183–189. doi:10.1016/j.jnoncrysol.2015.05.031
46. Santos SM, Cecilia JA, Vilarrasa-Garcia E, Junior IJS, Rodríguez-Castellón E, Azevedo DC. The effect of structure modifying agents in the SBA-15 for its application in the biomolecules adsorption. Microporous Mesoporous Mater. 2016;232:53–64. doi:10.1016/j.micromeso.2016.06.010
47. Wei J, Zou L. Synthesis of magnetical microspheres with tunable large pore mesostructures. J Porous Mater. 2016;23(3):577–581. doi:10.1007/s10934-015-0114-7
48. Ma B, Zhuang L, Chen S. Rapid synthesis of tunable-structured short-pore SBA-15 and its application on CO2 capture. J Porous Mater. 2016;23(2):529–537. doi:10.1007/s10934-015-0104-9
49. Xiong L, Du X, Shi B, Bi J, Kleitz F, Qiao SZ. Tunable stellate mesoporous silica nanoparticles for intracellular drug delivery. J Mater Chem B. 2015;3(8):1712–1721. doi:10.1039/C4TB01931A
50. Ma Y, Chen H, Shi Y, Yuan S. Low cost synthesis of mesoporous molecular sieve MCM-41 from wheat straw ash using CTAB as surfactant. Mater Res Bull. 2016;77:258–264. doi:10.1016/j.materresbull.2016.01.047
51. Wang J-G, Xiao Q, Zhou H-J, Sun P-C, Ding D-T, Chen T-H. Anionic surfactant-templated mesoporous silica (AMS) nano-spheres with radially oriented mesopores. J Colloid Interface Sci. 2008;323(2):332–337. doi:10.1016/j.jcis.2008.04.025
52. Xia Y, Mokaya R. Surfactant mediated control of pore size and morphology for molecularly ordered ethylene-bridged periodic mesoporous organosilica. J Phys Chem B. 2006;110(9):3889–3894. doi:10.1021/jp054513g
53. Vallet-Regí M, Balas F, Arcos D. Mesoporous materials for drug delivery. Angew Chem Int Ed. 2007;46(40):7548–7558. doi:10.1002/anie.200604488
54. Fang X, Zhao X, Fang W, Chen C, Zheng N. Self-templating synthesis of hollow mesoporous silica and their applications in catalysis and drug delivery. Nanoscale. 2013;5(6):2205–2218. doi:10.1039/C3NR34056F
55. Zhang K, Xu -L-L, Jiang J-G, et al. Facile large-scale synthesis of monodisperse mesoporous silica nanospheres with tunable pore structure. J Am Chem Soc. 2013;135(7):2427–2430. doi:10.1021/ja3116873
56. Bharti C, Nagaich U, Pal AK, Gulati N. Mesoporous silica nanoparticles in target drug delivery system: a review. Int J Pharm Invest. 2015;5(3):124–133. doi:10.4103/2230-973X.160844
57. Peng S, Huang B, Lin Y, Pei G, Zhang L. Effect of surface functionalization and pore structure type on the release performance of mesoporous silica nanoparticles. Microporous Mesoporous Mater. 2022;336:111862. doi:10.1016/j.micromeso.2022.111862
58. Yanagisawa T, Shimizu T, Kuroda K, Kato C. The preparation of alkyltrimethylammonium-kanemite complexes and their conversion to microporous materials. Bull Chem Soc Jpn. 1990;63(4):988–992. doi:10.1246/bcsj.63.988
59. ALOthman ZA. A review: fundamental aspects of silicate mesoporous materials. Materials. 2012;5(12):2874–2902. doi:10.3390/ma5122874
60. Gu D, Schüth F. Synthesis of non-siliceous mesoporous oxides. Chem Soc Rev. 2014;43(1):313–344. doi:10.1039/C3CS60155B
61. Jena BK, Raj CR. Electrochemical biosensor based on integrated assembly of dehydrogenase enzymes and gold nanoparticles. Anal Chem. 2006;78(18):6332–6339. doi:10.1021/ac060402r
62. Hu K, Lan D, Li X, Zhang S. Electrochemical DNA biosensor based on nanoporous gold electrode and multifunctional encoded DNA−Au bio bar codes. Anal Chem. 2008;80(23):9124–9130. doi:10.1021/ac8017197
63. Xu Z, Jiang X. Osteogenic TiO2 composite nano-porous arrays: a favorable platform based on titanium alloys applied in artificial implants. Colloids Surf A. 2022;640:128301. doi:10.1016/j.colsurfa.2022.128301
64. He Y, Li Z, Ding X, et al. Nanoporous titanium implant surface promotes osteogenesis by suppressing osteoclastogenesis via integrin β1/FAKpY397/MAPK pathway. Bioact Mater. 2022;8:109–123. doi:10.1016/j.bioactmat.2021.06.027
65. Yang J, Kang F, Wang X, Zhang Q. Design strategies for improving the crystallinity of covalent organic frameworks and conjugated polymers: a review. Mater Horiz. 2022;9(1):121–146. doi:10.1039/D1MH01170C
66. Xue M, Yang J, Kang F, Wang X, Zhang Q. Recent progress in single-crystal structures of organic polymers. J Mater Chem C. 2022;10(45):17027–17047. doi:10.1039/D2TC03907A
67. Côté AP, Benin AI, Ockwig NW, O’Keeffe M, Matzger AJ, Yaghi OM. Porous, crystalline, covalent organic frameworks. Science. 2005;310(5751):1166–1170. doi:10.1126/science.1120411
68. Li W-Y, Wan -J-J, Kan J-L, et al. A biodegradable covalent organic framework for synergistic tumor therapy. Chem Sci. 2023;14(6):1453–1460. doi:10.1039/D2SC06006J
69. Fang Q, Wang J, Gu S, et al. 3D porous crystalline polyimide covalent organic frameworks for drug delivery. J Am Chem Soc. 2015;137(26):8352–8355. doi:10.1021/jacs.5b04147
70. Bhanja P, Mishra S, Manna K, Mallick A, Das Saha K, Bhaumik A. Covalent organic framework material bearing phloroglucinol building units as a potent anticancer agent. ACS Appl Mater Interfaces. 2017;9(37):31411–31423. doi:10.1021/acsami.7b07343
71. Mousa AO, Lin Z-I, Chuang C-H, Chen C-K, Kuo S-W, Mohamed MG. Rational design of bifunctional microporous organic polymers containing anthracene and triphenylamine units for energy storage and biological applications. Int J Mol Sci. 2023;24(10):8966. doi:10.3390/ijms24108966
72. Palani P, Karpagam S. Conjugated polymers–a versatile platform for various photophysical, electrochemical and biomedical applications: a comprehensive review. New J Chem. 2021;45(41):19182–19209. doi:10.1039/D1NJ03803B
73. Lei Y, Zhu Z, Sun H, Mu P, Liang W, Li A. Conjugated microporous polymers bearing isocyanurate moiety as efficient antibacterial membrane and aerogels. Sep Purif Technol. 2020;248:117020. doi:10.1016/j.seppur.2020.117020
74. Wang Y, Yuan Q, Li M, Tang Y. Cationic conjugated microporous polymers coating for dual-modal antimicrobial inactivation with self-sterilization and reusability functions. Adv Funct Mater. 2023;33(19):2213440. doi:10.1002/adfm.202213440
75. Cheng S, Wang H, Pan X, et al. Dendritic hydrogels with robust inherent antibacterial properties for promoting bacteria-infected wound healing. ACS Appl Mater Interfaces. 2022;14(9):11144–11155. doi:10.1021/acsami.1c25018
76. Xu Q, He P, Wang J, et al. Antimicrobial activity of a conjugated polymer with cationic backbone. Dyes Pigm. 2019;160:519–523. doi:10.1016/j.dyepig.2018.08.045
77. Sun H, Chan W, Chen S, et al. Tissue paper-liked conjugated microporous polymers film for bacteria inhibition. J Environ Chem Eng. 2022;10(6):108933. doi:10.1016/j.jece.2022.108933
78. Mousa AO, Mohamed MG, Lin Z-I, Chuang C-H, Chen C-K, Kuo S-W. Conjugated microporous polymers as a novel generation of drug carriers: a systemic study toward efficient carriers of tetracycline antibiotic. Eur Polym J. 2023;196:112254. doi:10.1016/j.eurpolymj.2023.112254
79. Liu G, Bao S, Shi J, et al. Photoactive cationic conjugated microporous polymers containing pyrimidine with an ‘adsorption-kill’antibacterial strategy for infected wound healing. Chem Eng J. 2024;490:151593. doi:10.1016/j.cej.2024.151593
80. Davankov V, Rogoshin S, Tsyurupa M. Macronet isoporous gels through crosslinking of dissolved polystyrene. J Polym Sci Polym Symp. 1974;47:95–101. doi:10.1002/polc.5070470113
81. Davankov V, Timofeeva G, Ilyin M, Tsyurupa M. Formation of regular clusters through self-association of intramolecularly hypercrosslinked polystyrene-type nanosponges. J Polym Sci a Polym Chem. 1997;35(17):3847–3852. doi:10.1002/(SICI)1099-0518(199712)35:17
82. Tsyurupa M, Davankov V. Hypercrosslinked polymers: basic principle of preparing the new class of polymeric materials. React Funct Polym. 2002;53(2–3):193–203. doi:10.1016/S1381-5148(02)00173-6
83. Liao Q, Kim EJ, Tang Y, et al. Rational design of hyper-crosslinked polymers for biomedical applications. J Polym Sci. 2024;62(8):1517–1535. doi:10.1002/pol.20230843
84. Wang S, Li H, Huang H, Cao X, Chen X, Cao D. Porous organic polymers as a platform for sensing applications. Chem Soc Rev. 2022;51(6):2031–2080. doi:10.1039/D1CS00316B
85. Singh N, Son S, An J, et al. Nanoscale porous organic polymers for drug delivery and advanced cancer theranostics. Chem Soc Rev. 2021;50(23):12883–12896. doi:10.1039/D1CS00559F
86. Tang Y, Varyambath A, Ding Y, et al. Porous organic polymers for drug delivery: hierarchical pore structures, variable morphologies, and biological properties. Biomater Sci. 2022;10(19):5369–5390. doi:10.1039/D2BM00905D
87. Razzaque S, Guo L, Weng J, Su L, Tan B. Facile fabrication of hypercrosslinked microporous polymer nanospheres for effective inhibition of triple negative breast cancer cells proliferation. J Colloid Interface Sci. 2022;620:94–106. doi:10.1016/j.jcis.2022.03.109
88. Zhang T, Yu Y, Lu Y, et al. Bridging biodegradable metals and biodegradable polymers: a comprehensive review of biodegradable metal-organic frameworks for biomedical application. Prog Mater Sci. 2026;155. 101526. doi:10.1016/j.pmatsci.2024.101526
89. Deng Y, Guo M, Zhou L, et al. Prospects, advances and biological applications of MOF-based platform for the treatment of lung cancer. Biomater Sci. 2024;12(15):3725–3744. doi:10.1039/D4BM00379F
90. Giménez-Marqués M, Hidalgo T, Serre C, Horcajada P. Nanostructured metal-organic frameworks and their bio-related applications. Coord Chem Rev. 2016;307:342–360. doi:10.1016/j.ccr.2015.08.008
91. Bigham A, Islami N, Khosravi A, Zarepour A, Iravani S, Zarrabi A. MOFs and MOF-based composites as next-generation materials for wound healing and dressings. Small. 2024;20(30):2311903. doi:10.1002/smll.202311903
92. Chen X, Argandona SM, Melle F, Rampal N, Fairen-Jimenez D. Advances in surface functionalization of next-generation metal-organic frameworks for biomedical applications: design, strategies, and prospects. Chem. 2024;10(2):504–543. doi:10.1016/j.chempr.2023.12.015
93. Zeng Y, Yuan J, Ran Z, et al. Chitosan/NH2-MIL-125 (Ti) scaffold loaded with doxorubicin for postoperative bone tumor clearance and osteogenesis: an in vitro study. Int J Biol Macromol. 2024;263. 130368. doi:10.1016/j.ijbiomac.2024.130368
94. Zheng Z, Chen Y, Guo B, et al. Magnesium-organic framework-based stimuli-responsive systems that optimize the bone microenvironment for enhanced bone regeneration. Chem Eng J. 2020;396:125241. doi:10.1016/j.cej.2020.125241
95. Basharat M, Abbas Y, Hadji D, et al. Amorphous covalent inorganic-organic hybrid frameworks (CIOFs) with an aggregation induced selective response to UV-Visible light and their DFT studies. J Mater Chem C. 2020;8(39):13612–13620. doi:10.1039/D0TC03223F
96. Abbas Y, Ali S, Ali S, et al. Cyclotriphosphazene (P3N3) derived FeOx@SPNO-C core-shell nanospheres as peroxymonosulfate activator for degradation via non-radical pathway. Appl Surf Sci. 2024;645:158836. doi:10.1016/j.apsusc.2023.158836
97. Chen X, Li Z, Zhao H, et al. Enhancing maize yield and nutrient utilization through improved soil quality under reduced fertilizer use: the efficacy of organic-inorganic compound fertilizer. Agriculture. 2024;14(9):1482. doi:10.3390/agriculture14091482
98. Yang P, Gai S, Lin J. Functionalized mesoporous silica materials for controlled drug delivery. Chem Soc Rev. 2012;41(9):3679–3698. doi:10.1039/C2CS15308D
99. Estevão BM, Miletto I, Hioka N, Marchese L, Gianotti E. Mesoporous silica nanoparticles functionalized with amino groups for biomedical applications. ChemistryOpen. 2021;10(12):1251–1259. doi:10.1002/open.202100271
100. Vivero-Escoto JL, Slowing II II, Lin VS-Y. Tuning the cellular uptake and cytotoxicity properties of oligonucleotide intercalator-functionalized mesoporous silica nanoparticles with human cervical cancer cells HeLa. Biomaterials. 2010;31(6):1325–1333. doi:10.1016/j.biomaterials.2009.11.009
101. Chen X, Zhang Y, Arora P, Guan X. Nanopore stochastic sensing based on non-covalent interactions. Anal Chem. 2021;93(31):10974–10981. doi:10.1021/acs.analchem.1c02102
102. Fanourgakis GS, Gkagkas K, Tylianakis E, Froudakis G. A generic machine learning algorithm for the prediction of gas adsorption in nanoporous materials. J Phys Chem C. 2020;124(13):7117–7126. doi:10.1021/acs.jpcc.9b10766
103. Corriu R, Mehdi A, Reyé C. Nanoporous materials: a good opportunity for nanosciences. J Organomet Chem. 2004;689(24):4437–4450. doi:10.1016/j.jorganchem.2004.07.064
104. Kyriakides TR, Raj A, Tseng TH, et al. Biocompatibility of nanomaterials and their immunological properties. Biomed Mater. 2021;16(4):042005. doi:10.1088/1748-605X/abe5fa
105. Polarz S, Smarsly B. Nanoporous materials. J Nanosci Nanotechnol. 2002;2(6):581–612. doi:10.1166/jnn.2002.151
106. Yotsova R, Parushev I, Peev S, Dikova T. Potential applications of calcium pyrophosphate for bone regeneration: a systematic review. Cureus. 2025;17(4):e59123. doi:10.7759/cureus.59123
107. Yan Y, Gause KT, Kamphuis MMJ, et al. Differential roles of the protein Corona in the cellular uptake of nanoporous polymer particles by monocyte and macrophage cell lines. ACS Nano. 2013;7(12):10960–10970. doi:10.1021/nn404481f
108. Aramesh M, Yu D, Essand M, Persson C. Enhanced cellular uptake through nanotopography-induced macropinocytosis. Adv Funct Mater. 2024;34(28):2400487. doi:10.1002/adfm.202400487
109. Popova M, Koseva N, Trendafilova I, et al. Design of PEG-modified magnetic nanoporous silica based miltefosine delivery system: experimental and theoretical approaches. Microporous Mesoporous Mater. 2021;310:110664. doi:10.1016/j.micromeso.2020.110664
110. Bružauskaitė I, Bironaitė D, Bagdonas E, Bernotienė E. Scaffolds and cells for tissue regeneration: different scaffold pore sizes-different cell effects. Cytotechnology. 2016;68(3):355–369. doi:10.1007/s10616-014-9797-x
111. Norman JJ, Desai TA.Micro and nano engineered extracellular matrices.Silva GA, Parpura V;editors;Nanotechnology for Biology and Medicine: at the Building Block Level.Springer.Vol. 2011;101–121. doi:10.1007/978-1-4419-7853-0_6
112. Kim C, Lee JW, Heo JH, et al. Natural bone-mimicking nanopore-incorporated hydroxyapatite scaffolds for enhanced bone tissue regeneration. Biomater Res. 2022;26(1):7. doi:10.1186/s40824-022-00253-x
113. Shi M, Xia L, Chen Z, et al. Europium-doped mesoporous silica nanosphere as an immune-modulating osteogenesis/angiogenesis agent. Biomaterials. 2017;144:176–187. doi:10.1016/j.biomaterials.2017.08.027
114. Tabish TA, Pranjol MZI, Jabeen F, et al. Investigation into the toxic effects of graphene nanopores on lung cancer cells and biological tissues. Appl Mater Today. 2018;12:389–401. doi:10.1016/j.apmt.2018.07.004
115. Harish V, Tewari D, Gaur M, et al. Review on nanoparticles and nanostructured materials: bioimaging, biosensing, drug delivery, tissue engineering, antimicrobial, and agro-food applications. Nanomaterials. 2022;12(3):457. doi:10.3390/nano12030457
116. Duan F, Wang J, Li Z, Zhang T, Li Z, Zhou X. pH-responsive metal-organic framework-coated mesoporous silica nanoparticles for immunotherapy. ACS Appl Nano Mater. 2021;4(12):13398–13404. doi:10.1021/acsanm.1c02908
117. Wang Y, Shahi PK, Wang X, et al. In vivo targeted delivery of nucleic acids and CRISPR genome editors enabled by GSH-responsive silica nanoparticles. J Control Release. 2021;336:296–309. doi:10.1016/j.jconrel.2021.06.036
118. Yang W, Wang X, Ge Z, Yu H. Magnetically controlled millipede inspired soft robot for releasing drugs on target area in stomach. IEEE Robot Autom Lett. 2024;9(4):3846–3853. doi:10.1109/LRA.2024.3372467
119. Yang W, Cai S, Chen Y, et al. Modular and customized fabrication of 3D functional microgels for bottom-up tissue engineering and drug screening. Adv Mater Technol. 2020;5(5):1900847. doi:10.1002/admt.201900847
120. Athanassiadis AG, Ma Z, Moreno-Gomez N, et al. Ultrasound-responsive systems as components for smart materials. Chem Rev. 2022;122(5):5165–5208. doi:10.1021/acs.chemrev.1c00622
121. Teng X, Yu S, Qiao Z, Ge Z, Yang W. Dual-responsive tumbleweed-inspired soft robot based on poly(N‑isopropylacrylamide) and MoS2 for targeted drug delivery in stomach. J Bionic Eng. 2025;22(2):562–573. doi:10.1007/s42235-025-00650-7
122. Karmakar R, Dey S, Alam A, Khandelwal M, Pati F, Rengan AK. Attributes of nanomaterials and nanotopographies for improved bone tissue engineering and regeneration. ACS Appl Bio Mater. 2023;6(10):4020–4041. doi:10.1021/acsabm.3c00549
123. Lei L, Liu Z, Yuan P, et al. Injectable colloidal hydrogel with mesoporous silica nanoparticles for sustained co-release of microRNA-222 and aspirin to achieve innervated bone regeneration in rat mandibular defects. J Mater Chem B. 2019;7(16):2722–2735. doi:10.1039/C8TB03366K
124. Shyngys M, Ren J, Liang X, Miao J, Blocki A, Beyer S. Metal-organic framework (MOF)-based biomaterials for tissue engineering and regenerative medicine. Front Bioeng Biotechnol. 2021;9:603608. doi:10.3389/fbioe.2021.603608
125. Luo S, Sheng J, Jia X, Zhang G, Wang X, Hu J. Nitric oxide-releasing macromolecular systems in anticoagulant therapeutics. Macromol Chem Phys. 2023;224(23):2300196. doi:10.1002/macp.202300196
126. Harding JL, Reynolds MM. Metal organic frameworks as nitric oxide catalysts. J Am Chem Soc. 2012;134(7):3330–3333. doi:10.1021/ja210771m
127. Singh V. Theranostics: integrated diagnostics and therapy using nanomedicine. In: Nanomedicine: Innovations, Applications, and Breakthroughs in the Quest for Health and Medicine’s Future. Springer; 2024:505–530. doi:10.1007/978-3-031-57107-8_19
128. Ferreira CA, Goel S, Ehlerding EB, et al. Ultrasmall porous silica nanoparticles with enhanced pharmacokinetics for cancer theranostics. Nano Lett. 2021;21(11):4692–4699. doi:10.1021/acs.nanolett.1c00895
129. Chen Z, Sun Y, Wang J, et al. Dual-responsive triple-synergistic Fe-MOF for tumor theranostics. ACS Nano. 2023;17(10):9003–9013. doi:10.1021/acsnano.2c10310